Blog
Odio et unde deleniti. Deserunt numquam exercitationem. Officiis quo odio sint voluptas consequatur ut a odio voluptatem. Sit dolorum debitis veritatis natus dolores. Quasi ratione sint. Sit quaerat ipsum dolorem.

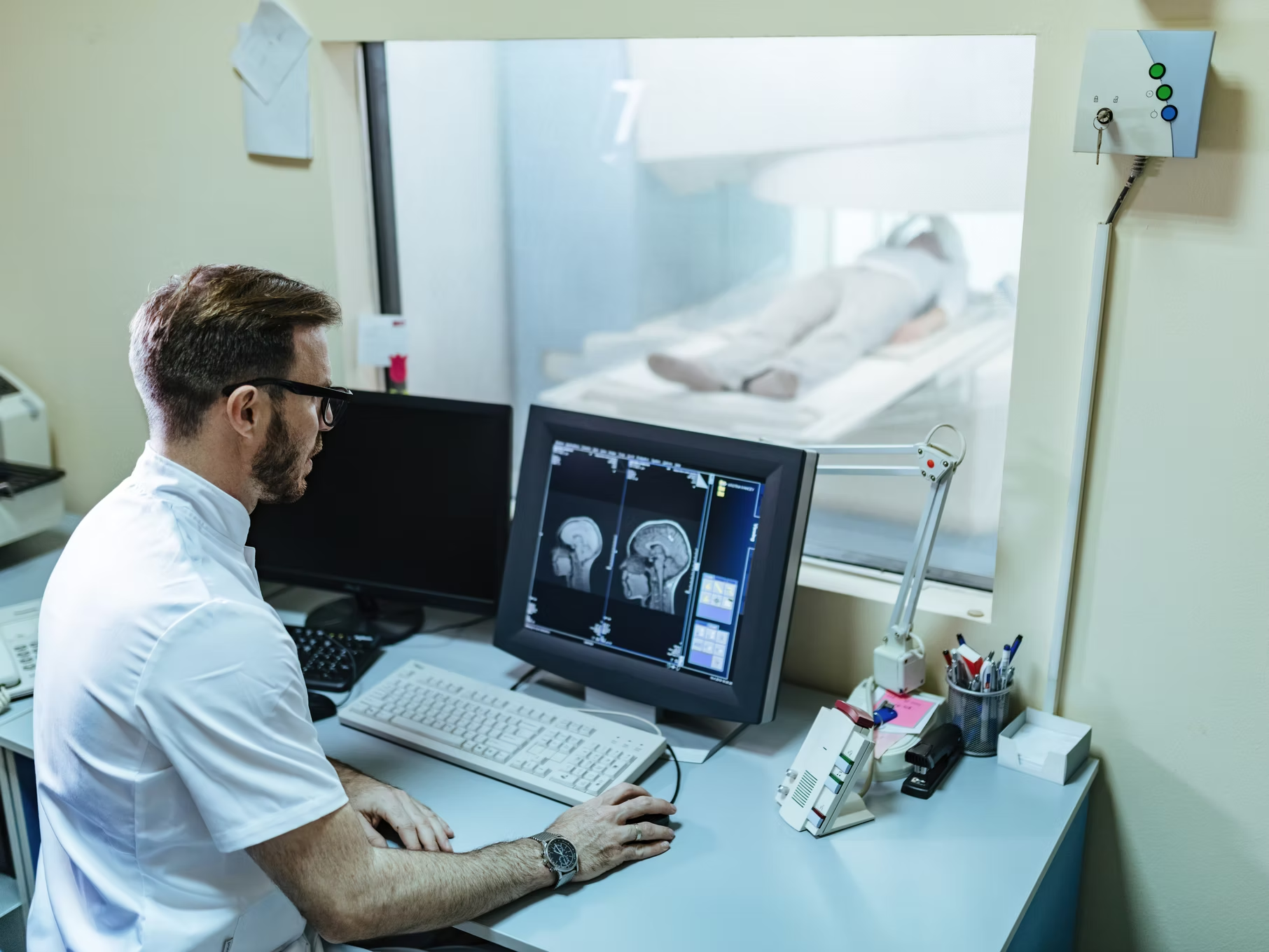
Head and neck cancer management
Feb 9, 2022 (14 min read)What are cancers of the head and neck? Cancers that are known collectively as head and neck cancers usually begin in the squamous cells...
 Dr.
Sampath
Chandra Prasad Rao
Dr.
Sampath
Chandra Prasad Rao
Head and neck cancer management
Although adenoids and tonsils are useful in protecting the body against bacteria and viruses, they have a minor role in immunity as age progresses. They tend to cause significant morbidity in children and adults with recurrent infections. Enlarged adenoids and tonsils can cause obstructive sleep apnea. Adenoids can lead to eustachian tube obstruction which leads to reduced hearing due to accumulation of fluid in the middle ear. Enlarged tonsils can also lead to accumulation of pus in the space behind the tonsils leading to sepsis and spread of infection elsewhere in the human body. The above-mentioned reasons are indications for removal of adenoids and tonsils.
The surgical procedures for removal of adenoids and tonsils are termed as adenoidectomy and tonsillectomy respectively. Adenoidectomy was previously a blind procedure which used to be carried out through the oral cavity with a curette. In the current era, adenoids are removed under vision with the help of an endoscope. The advent of gadgets like microdebrider and coablator has enabled the ENT surgeons to render the blood loss negligible. The removal of tonsils can be performed similarly with the gadgets mentioned above. In addition , LASER can be used for removal of the tonsils. Both adenoidectomy and tonsillectomy surgeries are performed without external incisions or scars. Complications of adenoid surgery although negligible include intra- and post-operative bleeding, injury to the Eustachian tube, velopharyngeal insufficieny and Grisels syndrome. Complications of tonsil surgery although negligible include intra- and post-operative bleeding and injury to neighbouring structures like soft palate, uvula, tongue and teeth.



Importance of early detection in Pediatric Hearing Loss
Jul 1, 2024 (9 min read)Importance of early detection in Pediatric Hearing Loss
 Dr.
Sandhya
PM
Dr.
Sandhya
PM
Importance of early detection in Pediatric Hearing Loss
360 million people (approximately 5% of the world’s population) live with disabling hearing loss and nearly million of them are children. It is estimated that over 60% of such hearing loss could be avoided through preventive measures. In addition, children who have hearing loss can benefit greatly from early identification and appropriate interventions.
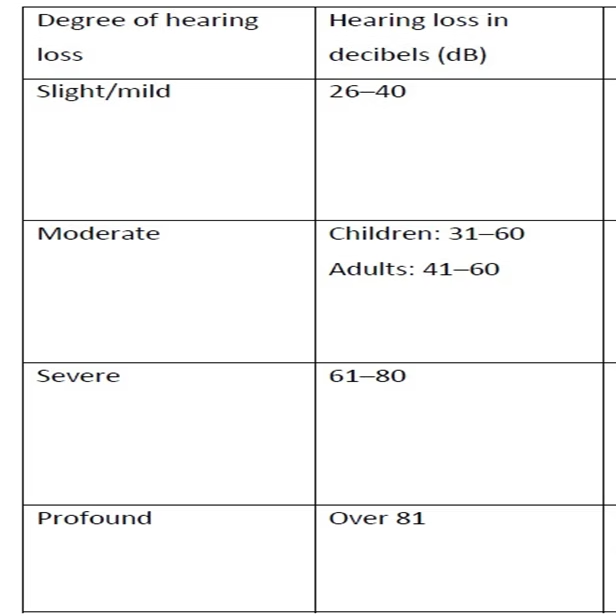
Disabling hearing loss in children is defined as hearing loss greater than 30 dB in the better-hearing ear.
The severity of the impact of hearing loss for a child depends on a number of factors:
. Age of onset: Children develop language in the early years of life. The impact of hearing loss on the development of spoken language is greatest in those who are born with hearing loss or develop it soon after birth. Degree of hearing loss: Hearing loss may range from mild to profound. The greater the severity, the greater the impact .
Different degrees of hearing loss.
. Age of identification and intervention:The sooner a child is identified
as
having hearing loss and the earlier he or she receives support, the
greater
the possibility of the child learning spoken language and the lower the
likely adverse impact of the hearing loss . The Joint Committee on
Infant
Hearing recommends that all children with hearing loss should receive
intervention by six months of age at the latest.
. Environment: The overall living environment, including access to
services,
significantly influences the development of a child with hearing loss.
Children with access to hearing technology, special education and sign
language may be able to participate at school and in social activities
“Hard of hearing” is people with hearing loss ranging from mild to
severe.
sounds (such as speech) are heard but not clearly understood. Such
people
usually communicate through spoken language and may benefit from hearing
amplification with hearing aids and cochlear implants . Deaf children
are
those with severe or profound hearing loss, which implies very little or
no
hearing. Hearing devices, such as cochlear implants, may help them to
hear
and learn speech.
The American Academy of Pediatrics Bright Futures EHDI prevention and
health
promotion program recommends formal hearing screening at age 4 years and
a
risk assessment for hearing loss at every well-child check between ages
1
and 4 years by asking parents about concerns for hearing loss to
identify
children who were lost to follow-up .
Timing of Hearing Loss
Congenital: identified in the neonatal period Delayed- onset: identified after the neonatal period but attributed to etiologies present at birth Acquired: occurs after the neonatal period and is attributed to etiologies not present at birth Sensorineural hearing loss: due to injury or defect within the cochlea, cochlear nerve, or the brainstem pathways to the auditory cortex Conductive hearing loss: due to injury or defect within the external or middle ear, including the external auditory canal, tympanic membrane, middle ear cavity, and ossicles Mixed hearing loss: combination of sensorineural and conductive types of hearing loss
Technology Used to Perform Screening
Otoacoustic Emissions (OAEs)
Sounds produced by outer hair cells in the cochlea in response to acoustic signals in the ear; this noninvasive test has different forms, known as transient evoked and distortion product OAE.
Auditory Brainstem Response (ABR)
A noninvasive test of the integrity of the auditory pathway from middle ear, to cochlea, to the vestibulocochlear nerve, and to brainstem, where the response is measured; the ABR can be used as a pass/fail test for screening or (softest sound) at which sounds are heard.
Automated Auditory Brainstem Response (AABR)
The hearing screening version of ABR for infants in the neonatal intensive care unit (NICU).
Protocols
Normal Newborn Nursery Hearing screening of full-term newborns usually involves the OAE.
If the infant does not pass the screen in one or both ears (termed as refer for diagnostic testing), then they may undergo a second screening at their primary care provider visitor the birth hospital before the age of 1month; a second screening refer should then cause the infant to undergo diagnostic ABR testing before the age of 3months. Neonatal Intensive Care Unit Hearing screening of infants from the NICU usually involves the AABR; a refer usually results in a transitory evoked otoacoustic emission test to rule out auditory neuropathy spectrum disorder and a diagnostic ABR before the age of 3 months (corrected for gestational prematurity).
Diagnosis of hearing disorders in early childhood
Diagnosis of hearing disorders in newborns and infants is generally a two-stage process. As described above, the current standard is UNHS, followed immediately by confirmatory diagnostic evaluation as appropriate.
Universal newborn hearing screening
The various studies on UNHS have either measured otoacoustic emissions (OAE) or performed automated auditory brainstem response (AABR) audiometry, or both. In two-stage screening, OAE measurement is followed by AABR audiometry.
Epidemiology
The prevalence of permanent bilateral severe to profound hearing loss in newborns is1.1per 1000 newborns.1 to 2 per 1000 newborns have bilateral mild to moderate hearing loss or unilateral hearing loss of any degree
Early identification allows for early interventions with parent-child programs, with a benchmark of no later than 3 to 6 months of age established by the Joint Committee on Infant Hearing, including hearing aids and intensive speech-language therapy,leads to better outcomes, including earlier integration into general education (i.e., mainstream schooling).
In addition to identifying infants with profound bilateral hearing loss, the newborn hearing screening programs also identify infants with bilateral mild to moderate or unilateral hearing loss.
Etiology of hearing loss:
Genetic factors. These are responsible for nearly 40% of childhood hearing loss. Hearing loss is much more frequent in children born of a consanguineous marriage.
Infections:
Perinatal -Rubella or cytomegalovirus. Childhood infections, such as meningitis, mumps and measles, can also cause hearing loss. Ear infections – e.g. chronic suppurative otitis media (CSOM)OME, can also lead to life-threatening problems, such as meningitis and brain abscesses .
Perinatal conditions:
Conditions at the time of birth can also lead to hearing loss like, Prematurity, low birthweight, birth asphyxia, neonatal jaundice, congenital malformations of the ear and the auditory nerve
Noise Exposure to loud sounds: Short, high intensity sounds, such as fireworks and shooting, may cause permanent hearing loss. The noisy machinery in a neonatal intensive care unit can also contribute to hearing loss
Medicines: Medicines, such as those used in the treatment of neonatal infections, malaria, drug-resistant tuberculosis and cancers, can lead to permanent hearing loss (ototoxic medicines).
WHO estimates that about 60% of hearing loss is due to preventable
causes:
Over 30% of childhood hearing loss is caused by infections, such as
rubella,
cytomegalovirus, mumps, meningitis, measles and chronic ear infections.
Meningitis and rubella together are responsible for over 19% of
childhood
hearing loss. Most of these infections can be prevented by immunization
and
good hygiene. Ear infections and glue ear can be prevented through good
ear
care and general hygiene, and can be treated by prompt medical and
surgical
interventions.
Complications at birth, such as lack of oxygen, low birthweight,
prematurity
and jaundice, account for 17% of childhood hearing loss. Such
complications
can be prevented through improved maternal and child health practices.
Use of ototoxic medicines in pregnant women and children is responsible
for
4% of childhood hearing loss, which could potentially be avoided.
The proportion of hearing loss due to preventable causes is much higher
in
middle- and lower-middle-income countries (75%) than in high-income
areas
(49%). The difference is probably explained by the overall higher
occurrence
of infections in the middle- and lower-middle-income countries and the
better maternal and child health care in high-income countries.
Early identification helps
Early identification of hearing loss needs to be followed by timely and appropriate interventions, in order to minimize developmental delays and promote communication, education and social development. The choice of interventions depends on the degree and the cause of hearing loss. Otitis media can often be treated and reversed by medical or surgical means . Hearing loss due to other causes cannot be reversed. However, its impact can be reduced through timely use of various approaches .
· hearing devices, such as hearing aids, and cochlear or middle ear implants; · hearing assistive technology, such as FM/radio systems and loop systems; · therapy to develop spoken language, such as auditory-verbal therapy, cued speech and auditory-oral therapy. · development of nonverbal communication, such as sign language.
Effects of hearing loss in children Hearing loss is a well-known prominent risk for speech and language developmental delay. The provision of hearing aids and cochlear implants early in life has been demonstrated to help many children attain near-normal speech and language trajectories, as measured by growth curves using standardized language scores. Hearing loss has also been found to affect a child’s quality of life, particularly in the school and social domains, as well as behavior and behavioral disorders. Children may develop hearing loss at a later age. Regular preschool and school-based hearing screening can identify hearing loss soon after its onset, allowing its adverse impact to be limited . For interventions to be effective, they should be appropriate, timely, family-centred and undertaken through an interdisciplinary approach, which includes audiological, medical, therapeutic and pedagogical services.
Strategies for prevention and care
A. Strengthening relevant programmes and organizations
· Strengthen immunization programmes , to prevent many of the infections that lead to hearing loss, such as congenital rubella, meningitis, mumps and measles. Potentially, over 19% of childhood hearing loss could be avoided through immunization against rubella and meningitis. · Strengthen maternal and child health programmes to prevent low birthweight, prematurity, birth asphyxia, congenital cytomegalovirus infection, and neonatal jaundice. Action: improve maternal and neonatal care through 1. improved nutrition, 2. awareness of hygienic practices, 3. promotion of safe birth, 4. prompt management of neonatal infections and jaundice · Strengthen organizations of people with hearing loss, parents and family support groups.
B. Implementation of screening and intervention programmes
Implement newborn and infant hearing screening with tracking, and initiate appropriate interventions to identify and treat children with congenital or early-onset hearing loss . A newborn hearing screening programme should follow a family-centred approach, in which families are empowered to make decisions for their children . Action: put early intervention programmes in place and implement newborn hearing screening programmes (based on physiological methods) that focus on: · appropriate interventions, ideally initiated before 6 months of age; · family support, including guidance and counselling of parents; · hearing rehabilitation through hearing aids and cochlear implants; · suitable therapy and communication options. · Implement school-based hearing screening with the aim of identifying, referring and managing common ear diseases and hearing loss. Action: integrate ear and hearing screening in school health programmes and develop links for provision of suitable medical, surgical and rehabilitative care.
C. Training:
· Train primary-level physicians and health care workers about the relevance of ear diseases, the need for early intervention to address hearing loss and the available treatment options. This would allow provision of accessible services and facilitate referral for management of ear diseases and hearing loss. · Train otologists, audiology professionals, other medical professionals (such as nurses), therapists and teachers to provide the required care and services. This is an important step in addressing ear and hearing problems.
D. Technologies Making appropriate accessible:
Make hearing devices accessible. Advances in the field of hearing aids and cochlear implants have considerably improved the available options for people with hearing loss. Despite this, only a fraction of those who need these devices have access to them. Particularly in developing countries, there are several significant barriers to access to hearing aids for people with hearing loss. A major barrier is the cost of hearing aids, batteries and maintenance. There is also a scarcity of health care professionals able to fit, maintain and repair devices. Transportation costs and travel time to a health centre may be prohibitive for many people with hearing loss, especially in rural areas . Technological advances, such as solar-powered or self-fitting hearing aids, may help to overcome some of these significant barriers in the future. Action: develop sustainable initiatives for affordable fitting and maintenance of hearing devices, and provide ongoing support for people using these devices. Make communication and education accessible . A deaf child benefits greatly from early introduction to language. This may be in the form of rehabilitation for verbal communication (such as auditory-verbal and auditory-oral therapy). Policy-makers should also promote alternative means of communication, including sign language, total communication, bilingual/bicultural (bi-bi) teaching, cued speech and lipreading approaches . Use of loop and FM systems in classrooms and public places, as well as provision of captions on audiovisual media, are important for improving accessibility of communication for people with hearing .
E. Regulation and monitoring:
. Regulate and monitor the use of ototoxic medicines, in order to minimize the dangers posed by their indiscriminate use . Where their use is unavoidable, regular audiological monitoring will help identify hearing loss at an early stage. . Regulate and monitor noise levels in the community, especially at recreational venues and sports arenas. The addition of safety features to personal audio systems can help to reduce the risk of hearing loss associated with their use .
F. Raising awareness:
. Raise awareness about healthy ear care practices that can reduce ear infections . For instance, avoiding insertion or instillation of any substance into the ear can help to decrease ear problems. Ensuring that children with ear pain are not treated with home remedies and consult a medical practitioner can prevent chronic ear infections and the associated hearing loss. . Raise awareness about the dangers of loud sounds by educating children at an early age about the risks associated with high volumes, especially in a recreational context (firecrackers, loud music, use of headphones, noisy games) . This can help to modify behaviour patterns and promote safe listening, which in turn can prevent the development of noise-induced hearing loss during childhood and adolescence or later in life.

World Hearing Day 2024
Feb 20, 2024 (1 min read)World Hearing Day is held on 3 March each year to raise awareness on how to prevent deafness and hearing loss and promote ear and hearing...
World Hearing Day 2024
Hearing loss has often been referred to as an “invisible disability”, not just because of the lack of visible symptoms, but because it has long been stigmatized in communities and ignored by policy-makers.

A Comprehensive Guide to Facial Rehabilitation Protocol
Sep 9, 2023 (4 min read)What is facial rehabilitation? Facial rehabilitation protocol, often referred to as facial therapy or facial retraining, is a specialized...
 Dr
.Lavanya
Manoharan
Dr
.Lavanya
Manoharan
A Comprehensive Guide to Facial Rehabilitation Protocol
What is facial rehabilitation?
Facial rehabilitation protocol, often referred to as facial therapy or facial retraining, is a specialized area of rehabilitation that focuses on restoring and improving facial muscle function. This protocol is crucial for individuals who have experienced facial nerve damage, paralysis, or any condition that affects their facial expressions and functions. Whether due to injury, surgery, or a neurological disorder, facial rehabilitation can significantly enhance an individual's quality of life by helping them regain control of their facial muscles and expressions.
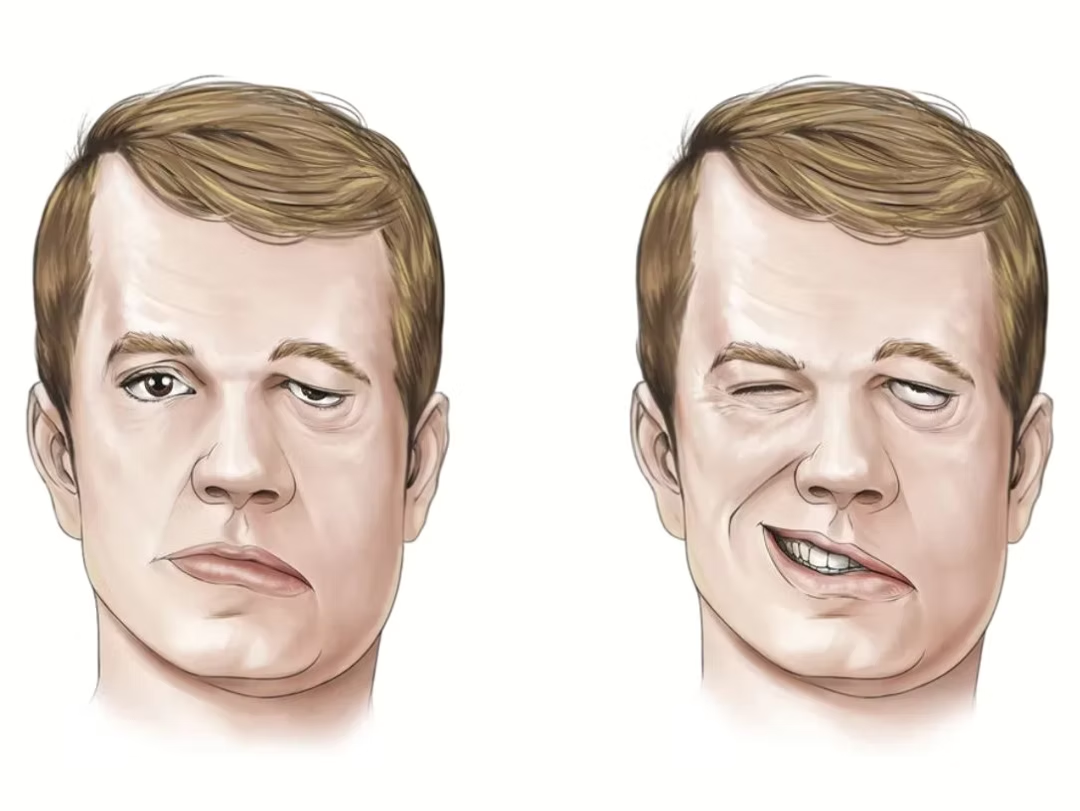
How to assess facial palsy?
Different scoring systems are available, of them the commonly used is the House-Brackmann scoring system. When considering the synkinesis component also the Sunnybrook and E-face system are more comprehensive. E -Face grading systems can also be used by the individuals themselves for monitoring the progress.

*Emerging vs Time-Tested Methods of Facial Grading Among Patients With Facial Paralysis
Robert A. Gaudin, Mara Robinson, Caroline A. Banks, Jennifer Baiungo, Nate Jowett, and Tessa A. Hadlock JAMA Facial Plastic Surgery 2016 18:4, 251-257.
What are the goals and techniques?
Facial rehabilitation is a multi-faceted approach that combines various techniques and exercises to address a wide range of issues affecting the face. These issues can include facial nerve palsy, Bell's palsy, facial muscle weakness, post-surgery recovery, and even facial muscle imbalances resulting from habits or lifestyle factors.
The primary goals of facial rehabilitation protocol are to:
1. Restore facial muscle strength: Strengthening exercises help
individuals
regain control over their facial muscles, enabling them to perform basic
facial movements such as smiling, frowning, and blinking.
2. Improve facial symmetry: Many individuals with facial nerve damage or
paralysis experience asymmetry in their facial features. Facial
rehabilitation aims to correct this imbalance and restore a more natural
appearance.
3. Enhance facial coordination: Coordinating facial muscle movements is
essential for a wide range of functions, from speaking and eating to
emotional expression. Facial therapy helps individuals regain control
and
coordination of these muscles.
4. Manage pain and discomfort: Some individuals may experience pain,
discomfort, or tightness in their facial muscles. Rehabilitation can
help
alleviate these symptoms and improve overall comfort.
5. Boost confidence and self-esteem: Restoring facial function and
appearance
can have a profound impact on an individual's self-esteem and
confidence.
Facial rehabilitation can help individuals feel more comfortable in
social
situations and improve their overall quality of life.
Facial rehabilitation protocol typically involves a combination of the following components:
1. Education: Understanding the underlying condition and the importance
of
adhering to the rehabilitation process is the first step. Patients learn
about their specific condition, the anatomy of facial muscles, and the
expected outcomes of rehabilitation.
2. Exercises: A tailored exercise regimen is a cornerstone of facial
rehabilitation. These exercises target specific muscle groups, helping
individuals regain control, strength, and coordination of their facial
muscles. Exercises can be passive (performed by a therapist) or active
(performed by the patient).
3. Massage and manual therapy: Gentle massage and manual techniques are
used
to
improve blood circulation, reduce muscle tension, and promote relaxation
in
the facial muscles.
4. Neuromuscular re-education: This technique focuses on retraining the
nerves
and muscles to work together effectively. It involves activities that
require conscious effort to control facial movements.
5. Biofeedback: Biofeedback technology can be used to provide patients
with
real-time information about their muscle activity, helping them learn
how to
control their facial muscles more effectively.
6. Electrical stimulation: In some cases, electrical stimulation may be
employed to stimulate facial muscles and promote muscle re-education.
7. Emotional support: Coping with the physical and emotional challenges
of
facial muscle dysfunction can be challenging. Emotional support from a
therapist or support group can be a vital component of the
rehabilitation
process.
It's important to note that facial rehabilitation protocols are highly individualized. Each patient's condition is unique, and their treatment plan should reflect that.
What are the surgical options in Facial rehabilitation?
Facial rehabilitation involves both static and dynamic surgical procedures to address a range of facial conditions. Static procedures focus on restoring facial aesthetics and structure while maintaining a relatively fixed appearance. These can include procedures like facelifts or facial implants. Dynamic surgical procedures, on the other hand, aim to restore facial movement and expression, often used in cases of facial paralysis. Techniques such as facial nerve grafts or muscle transfers enable patients to regain natural facial animation.
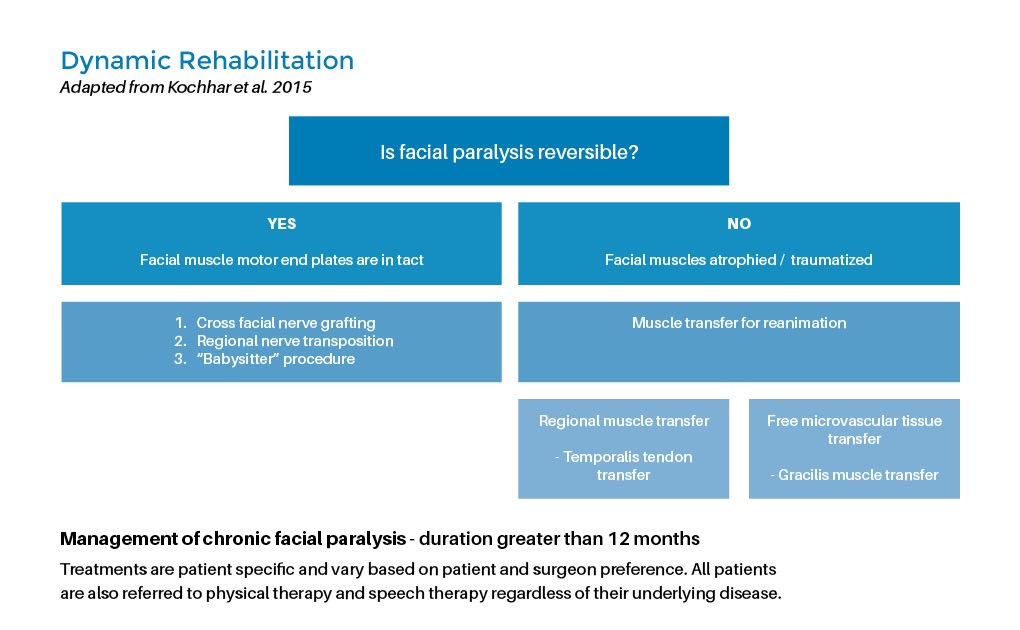
What is the duration of Facial Rehabilitation?
Duration of facial rehabilitation varies from person to person and depends on several factors, including the severity of the condition, the individual's commitment to the therapy, and their overall progress. Some individuals may see significant improvement within a few weeks, while others may require several months of therapy. Patients are typically encouraged to continue their exercises and maintenance routines even after their initial rehabilitation program is complete. This helps ensure long-term success and continued improvement in facial muscle function.
Conclusion:
Facial rehabilitation protocol is a vital and effective approach for individuals with facial muscle dysfunction. Whether it's due to nerve damage, paralysis, or other factors, this specialized rehabilitation can significantly improve facial function, appearance, and overall quality of life. If you or someone you know is dealing with facial muscle issues, consider seeking the expertise of a healthcare professional trained in facial rehabilitation at Bangalore head and neck sciences to create a tailored treatment plan and embark on the journey to facial recovery and rejuvenation.
Allergy Skin Prick Test
Aug 7, 2023 (3 min read)What is Allergy ? Allergy is a clinical state where the body responds in an exaggerated manner to foreign stimuli called Allergen....
 Dr.
Saud
Ahmed
Dr.
Saud
Ahmed
Allergy Skin Prick Test
What is Allergy ?
Allergy is a clinical state where the body responds in an exaggerated manner to foreign stimuli called Allergen. Allergy is known to run in families and has a genetic predisposition. After the initial exposure to an allergen, a genetic predisposed individual produces specific molecules called Ig E antibodies. During the subsequent exposure to a similar allergen, the Ig E antibodies cross link causing release of bioactive amine called histamine which is stored in mast cells. It is the histamine that causes most of the early symptoms of allergy.
What are the common Environmental Allergens ?
Pollen grains, House Dust mites, mold, insects, animal dander and certain food products are the common allergens. The Allergens could be perennial like dust mites and insects or could be seasonal as in pollen grains. Pollen is released during the flowering season and this explains the seasonal exacerbation of symptoms in individuals allergic to certain pollen grains.
What are the symptoms of Allergy ?
Depending on the organ involved the symptoms could range from frequent colds, cough and wheezing, itchy red eyes and itchy skin rashes. In some individuals severe allergy reaction can cause life threatening event called anaphylaxis.
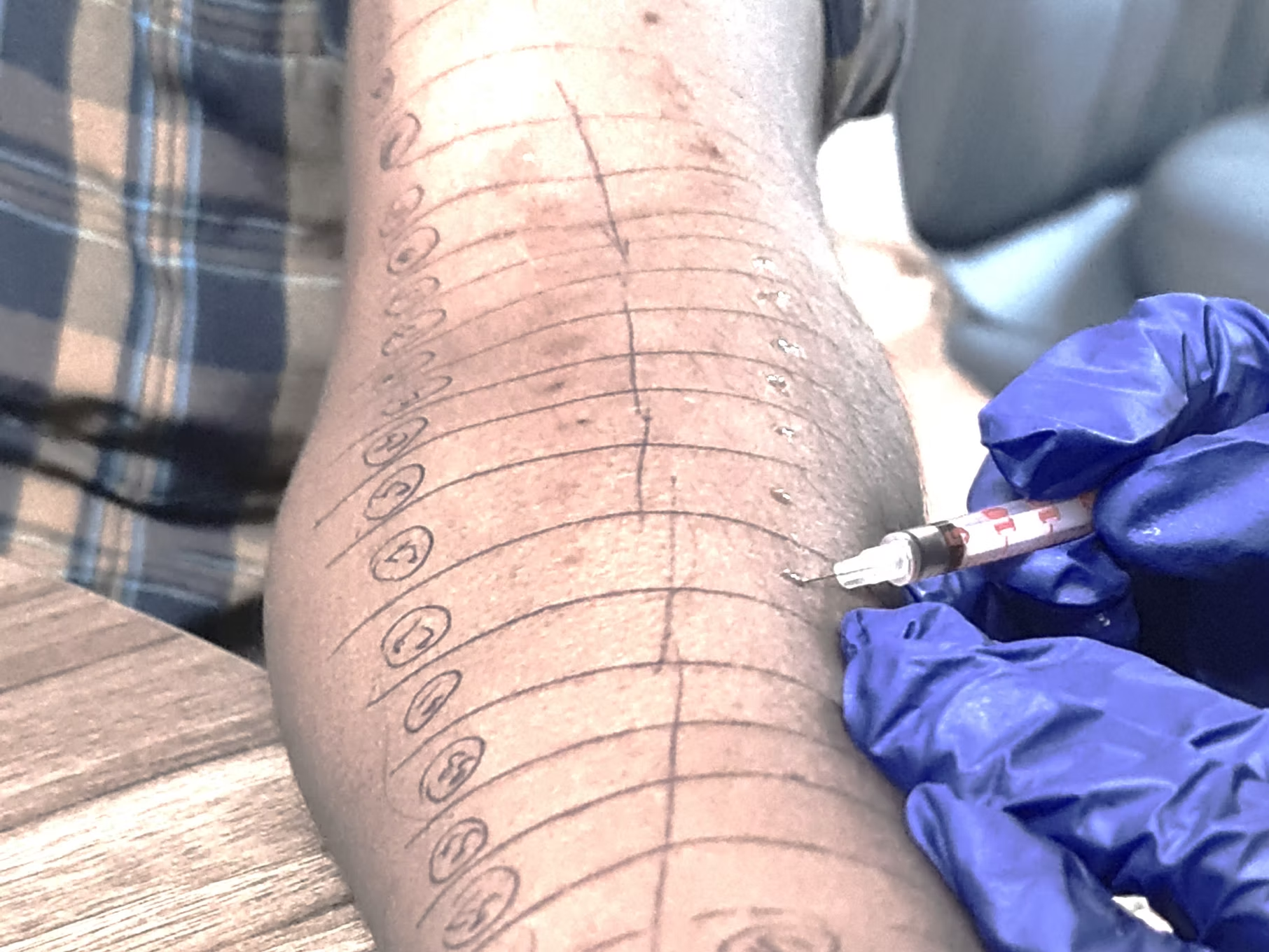
What is Allergy Skin Prick Test ?
This is a gold standard test to find out the causative agent for allergy. This is generally performed on individuals after 5 years of age with a strong suspicion of allergy. Once the severity of symptoms is brought down with medications, a skin prick test is scheduled. Care is taken to stop anti histamine medications a week prior to the test as these medications can interfere with the test results. Nasal sprays, drops and medicines like corticosteroids and monteleukast do not interfere with the test. However it is important that the individual being tested should furnish details of the all the current medications being taken including AYUSH related drugs. The test is a painless and bloodless procedure done on the forearm or back. A set of allergens are selected for the test. These are selected based on the nature of symptoms (whether perennial or seasonal) and geographical location of the patient. The allergen extracts are then placed drop by drop over the skin and the area is gently pricked with a tiny lancet. A positive and negative control testing with histamine and saline respectively is also performed in order to ensure there is no false positive or negative results. The results are interpreted after about 20 minutes of the prick. A reddish itchy swelling over the skin indicates a positive reaction. The size of the redness and swelling is measured and documented. The test results are obtained in the same sitting. Some amount of local skin itching and redness maybe observed after the test which is self-limiting.
Is the test safe ?
The test is safe and doesn’t cause any major problem. Special Precaution is taken before the test to make sure that the individuals being tested do not have any acute or critical ongoing allergy symptoms like severe wheezing. The center performing the test is well equipped with emergency medications and trained doctors to handle any untoward medical emergency arising from the procedure.
Who cannot undergo the test ?
Very small children, pregnant ladies and individuals with recent anaphylaxis are not tested. Those with acute allergy symptoms are first treated with allergy medicines and after becoming stable are tested. People who are on antihistamines or any other traditional / Alternative therapy should withhold medications before the test.
Why is testing important ?
It helps us identify the causative factor for allergy. Appropriate avoidance measures can be followed by the individual to reduce the exposure to the identified allergen. Allergen immunotherapy / Vaccine in the form of sub cutaneous injections or sublingual drops and tablets for allergy can also be offered based on the skin prick test result in order to provide long term benefit and cure from allergy. For more information on Allergy testing and immunotherapy, kindly get in touch with our specialist doctors at Bangalore ENT Institute.
Understanding CSF Rhinorrhea: Causes, Symptoms, and Treatment Options
Jul 10, 2023 (7 min read)What is CSF Rhinorrhea? Cerebrospinal fluid (CSF) rhinorrhea is a condition characterized by the leakage of cerebrospinal fluid from the...
 Dr.
Niveditha
Damodharan
Dr.
Niveditha
Damodharan
Allergy Skin Prick Test
Understanding CSF Rhinorrhea: Causes, Symptoms, and Treatment Options
What is CSF Rhinorrhea?
Cerebrospinal fluid (CSF) rhinorrhea is a condition characterized by the leakage of cerebrospinal fluid from the nose. CSF is the clear fluid that surrounds and protects the brain and spinal cord. When this fluid escapes through the nasal passages, it can indicate a potential problem in the protective barrier surrounding the central nervous system. This blog post will delve into the causes, symptoms, and treatment options for CSF rhinorrhea.
Why does it happen?
The most common cause of CSF rhinorrhea is trauma to the head or face, such as a severe blow or fracture to the skull base. This can create a tear or hole in the dura mater, the thick membrane that encases the brain and spinal cord, allowing CSF to leak into the nasal cavity. Other causes include:
1. Congenital Defects: Some individuals may be born with abnormalities in the skull base or the thin bone that separates the nasal cavity from the brain, increasing the risk of CSF leakage. In some cases, there can be openings or weak spots in the bone of the lateral part of the sphenoid sinus roof. These openings can happen because a canal called Sternberg's canal, which is normally present during embryonic development, doesn't close properly. When these openings occur, they can lead to CSF leaks and the development of eningoencephaloceles, which are abnormal protrusions of the brain tissue and meninges. 2. Surgical Complications: Certain surgical procedures, particularly those involving the sinuses or skull base, can inadvertently damage the dura mater and lead to CSF rhinorrhea. 3. Tumors: Tumors within the nose and paranasal sinuses or in the brain can cause CSF leak. Brain Tumors can exert pressure on the brain tissue, impair the normal flow of CSF, and raise intracranial pressure(ICP). This increased pressure can eventually result in CSF leakage through the nasal passages. Tumors or abnormal growths in the nasal or sinus cavities can erode the bone and create a passage for CSF to leak. 4. Hydrocephalus: Hydrocephalus is a condition characterized by the accumulation of excess CSF within the ventricles of the brain. As the ventricles enlarge, the pressure within the skull increases, potentially causing CSF to leak through the nose. 5. Intracranial Hypertension: Certain medical conditions, such as idiopathic intracranial hypertension (IIH) can cause elevated ICP without an apparent underlying cause. This increased pressure can contribute to CSF leakage.
What are the symptoms?
The primary symptom of CSF rhinorrhea is the persistent and watery discharge of fluid from one or both nostrils. The fluid may appear clear and taste salty, as it resembles CSF. Other symptoms may include:
1. Frequent or recurrent nasal infections
2. Headaches
3. Loss of smell or taste
4. Ringing in the ears (Pulsatile tinnitus)
5. Neck stiffness or pain
6. Visual disturbances, such as double vision or changes in vision
It's important to note that not all cases of clear nasal discharge indicate CSF rhinorrhea. Other causes, such as allergies or a common cold, can also produce similar symptoms. Therefore, a proper medical evaluation is essential for an accurate diagnosis.
How do we diagnose?
If CSF rhinorrhea is suspected, a thorough medical evaluation is necessary to confirm the diagnosis and determine the underlying cause. Diagnostic tests may include:
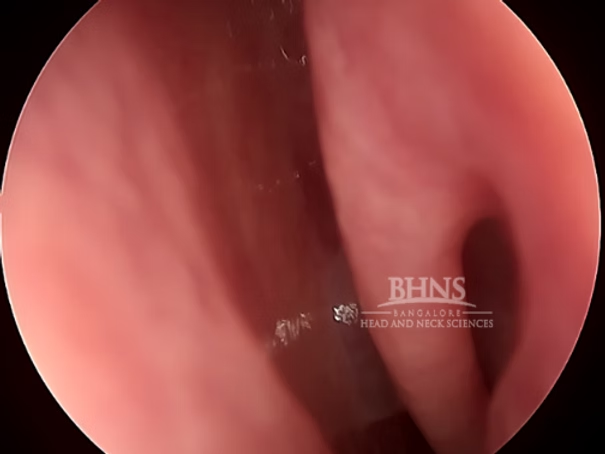
Physical Examination: An ENT surgeon will assess the nasal discharge, check for any signs of trauma or abnormalities, and evaluate neurological function. An outpatient procedure called endoscopy can also diagnose a CSF leak.
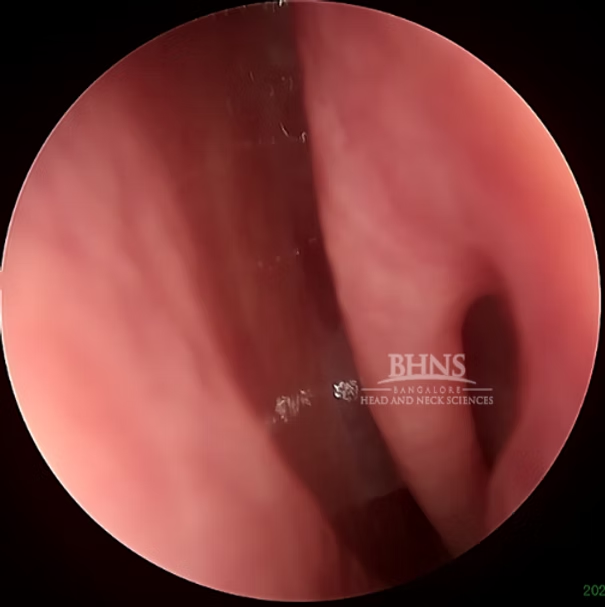
Imaging: Several imaging techniques are used to diagnose and evaluate CSF rhinorrhea. Here are some commonly employed imaging procedures:
High-Resolution CT Scan: This imaging technique provides detailed images of the skull base and surrounding structures. It can help identify fractures, bony abnormalities, or tumors that may be causing CSF leakage.
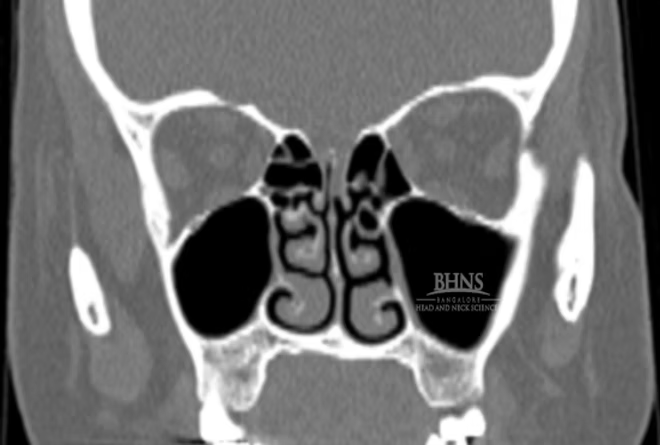
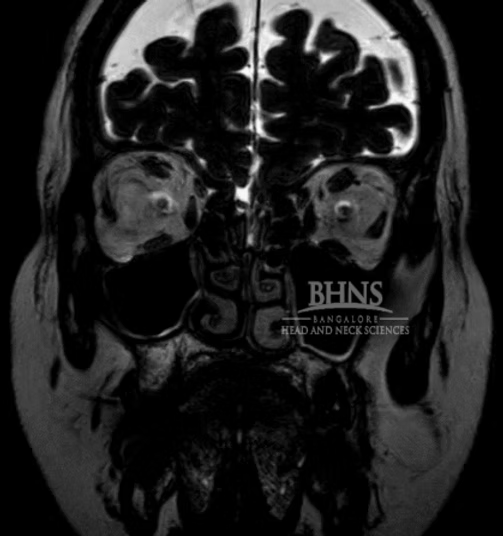
1. Intrathecal Fluorescein: In this procedure, a fluorescent dye called fluorescein is injected into the CSF through an injection given at the back. If there is a CSF leak, the dye can be visualized as it flows through the nasal passages, helping to identify the site of leakage. 2. Radionuclide Cisternogram: This imaging study involves injecting a small amount of radioactive material into the CSF. A specialized camera then tracks the movement of the radioactive tracer, allowing visualization of CSF flow and identification of any abnormal leakage points. 3. CT Cisternography: This technique combines a CT scan with the injection of a contrast dye into the CSF. It provides detailed images of the CSF pathways and can detect the presence of leaks or abnormalities. 4. MR and MR Cisternography: Magnetic resonance imaging (MRI) is used to obtain detailed images of the brain, skull base, and CSF spaces. MR Cisternography involves injecting a contrast agent into the CSF to enhance the visualization of CSF flow and identify areas of leakage or blockages.
These imaging techniques help in locating the site of CSF leakage, identifying underlying causes, and guiding appropriate treatment decisions.
. Laboratory Analysis: Analyzing the nasal discharge can help differentiate CSF from other types of fluids. When investigating CSF rhinorrhea, laboratory tests can be performed on the collected nasal discharge to aid in diagnosis. Here are some laboratory tests commonly used for CSF rhinorrhea:
a. Beta-2 Transferrin Assay: This highly specific test detects the presence of beta-2 transferrin, a protein found exclusively in CSF. It helps confirm the presence of CSF in nasal discharge, distinguishing it from other fluids. It has 100% sensitivity and 95% specificity. b. Beta Trace protein: Beta-trace protein (βTP) is another chemical marker that could be used for the detection of CSF. This is the second most common protein found in CSF after albumin. βTP has been identified as a prostaglandin D2 synthase. It is produced by the meninges and choroid plexus and released into CSF. This protein is also present in other body fluids, including serum, but at much lower concentrations than in CSF. Detection of βTP has 100% sensitivity and specificity in cases of confirmed CSF rhinorrhea. c. Glucose Levels: CSF glucose levels are measured to assess the metabolic status of the central nervous system. Abnormal glucose levels may indicate certain underlying conditions like infections. d. Protein Levels: The measurement of protein levels in the CSF can provide insights into the presence of inflammation, infections, or other abnormalities. e. Cell Count and Differential: This test examines the number and types of cells present in the CSF. An increased number of white blood cells may suggest an infection or inflammation. f. Gram Stain and Culture: These tests involve staining and culturing the CSF sample to identify bacterial pathogens responsible for any associated infections.
Are there any home-based tests to distinguish cerebrospinal fluid (CSF) from other types of nasal discharge?
Certainly! One home-based method is the handkerchief test. It involves using a handkerchief to collect the discharge from the nose. If the discharge is CSF, it will not become stiff or rigid when absorbed by the handkerchief. However, if the discharge is from the nose, it will stiffen due to the presence of mucin. This test can provide some initial observations, but it's important to remember that a proper medical evaluation by a healthcare professional is necessary for an accurate diagnosis of CSF rhinorrhea.
How to treat CSF Rhinorrhea?
Treatment options for CSF rhinorrhea depend on the underlying cause, location, and severity of the leakage. Conservative measures may include bed rest, strict avoidance of activities that increase intracranial pressure (e.g., heavy lifting, straining), and antibiotics to prevent or treat infections. Surgical intervention is often necessary to repair the tear or defect in the dura mater and prevent further CSF leakage. Several techniques may be employed, including:
. Endoscopic Repair: Minimally invasive procedures that use specialized endoscopic instruments and cameras to visualize and repair the defect in the dura, typically with the assistance of additional techniques such as grafts or sealants. The location of the CSF leak is determined, and the hole in the dura mater is enlarged until the bony edge of the skull base becomes visible. To address any accompanying meningoencephaloceles, techniques such as bipolar cautery or coblation may be employed to destroy the abnormal tissue. It is important to note that the meningoencephalocele should not be pushed back into the skull without proper treatment. Once the bony rim of the defect is identified, the nasal mucosa surrounding the hole is removed. This allows for proper adhesion of any graft to the bone, resulting in an improved seal to prevent further CSF leakage. We use grafts like fat, muscle, cartilage or bone to seal the defect. Local vascular flaps like nasoseptal flap, mucosal flap from turbinates or free fascia can be used to secure the defect. The grafts are einforced with the help of materials such as Surgicel, Gel foam and Tissue glue.
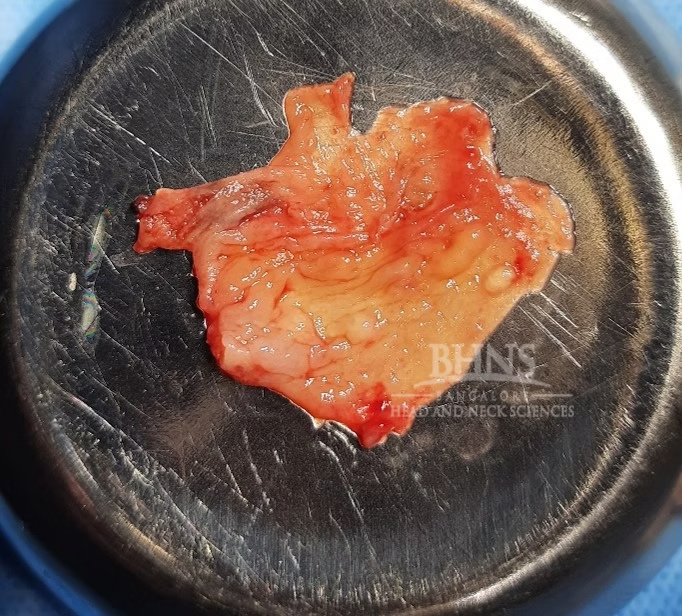
. Open-Surgical Repair: Open-surgical approaches are sometimes required to effectively close a CSF leak. Here are a few common open approaches used for this purpose:
a. Craniotomy: In this procedure, a section of the skull is removed to gain direct access to the area of CSF leakage. The dural defect is repaired, and any accompanying meningoencephalocele or other abnormalities are addressed. Afterwards, the removed section of the skull is usually replaced and secured back into position. b. Transcranial Approaches: These approaches involve accessing the site of the CSF leak through specific routes within the cranium, such as the frontal or temporal regions. The dural defect is repaired directly, and any associated abnormalities are treated. c. Combined Approaches: In some complex cases, a combination of surgical approaches may be necessary to adequately address the CSF leak. This may involve a combination of endoscopic-assisted techniques and transcranial approaches, depending on the location and extent of the defect.
It's important to note that the choice of approach depends on several factors, including the location and size of the CSF leak, associated complications, and the surgeon's expertise. The ultimate goal of these surgical approaches is to repair the dural defect, restore the normal flow of CSF, and prevent further leakage.
Postoperative care typically involves close monitoring, restricted physical activity, and follow-up examinations to ensure the successful closure of the CSF leak. Medications to reduce CSF production can be added in cases of high-pressure leaks associated with IIH. We do not recommend lumbar drains as a routine at our centre.
Take home message!
CSF rhinorrhea is a condition characterized by the leakage of cerebrospinal fluid from the nose, typically due to trauma, congenital defects, or tumors. It is important to recognize the symptoms and seek medical attention for a proper diagnosis and treatment. Early intervention is crucial to prevent complications such as infections and meningitis. If you suspect CSF rhinorrhea or have persistent nasal discharge, consult our doctors at Bangalore Skull Base Institute to undergo the necessary evaluations and receive appropriate care.
Anomalies of Cochlea & their Management
Jan 30, 2023 (3 min read)Human ear consists of the outer, middle and inner ear and Inner ear complex that participates in Sensorineural hearing is the Cochlea....
 Dr.
Rakshita Kamath
Dr.
Rakshita Kamath
Anomalies of Cochlea & their Management

Understanding CSF Rhinorrhea: Causes, Symptoms, and Treatment Options
Human ear consists of the outer, middle and inner ear and Inner ear complex that participates in Sensorineural hearing is the Cochlea. Birth related abnormalities in development of cochlea are called Anomalies/malformations of the Cochlea. Malformed inner ear complex are caused by interruption during various parts of development of the ear during the first 3 months of foetal development. They are the predominant causes of Sensorineural related hearing loss.

These malformations represent about 20% of all patients with sensorineural hearing loss. (1) From the first reported case in 1791 in Italy by Carlo Mondini, there has only been continual evolution in the world of inner ear impairments being discovered and discussions on their management have evolved in the last 230yrs. (2,3)
Sennaroglu and colleagues have extensively worked in this regard and the most accepted classification that is currently being followed in centres worldwide was given in 2017.(4)
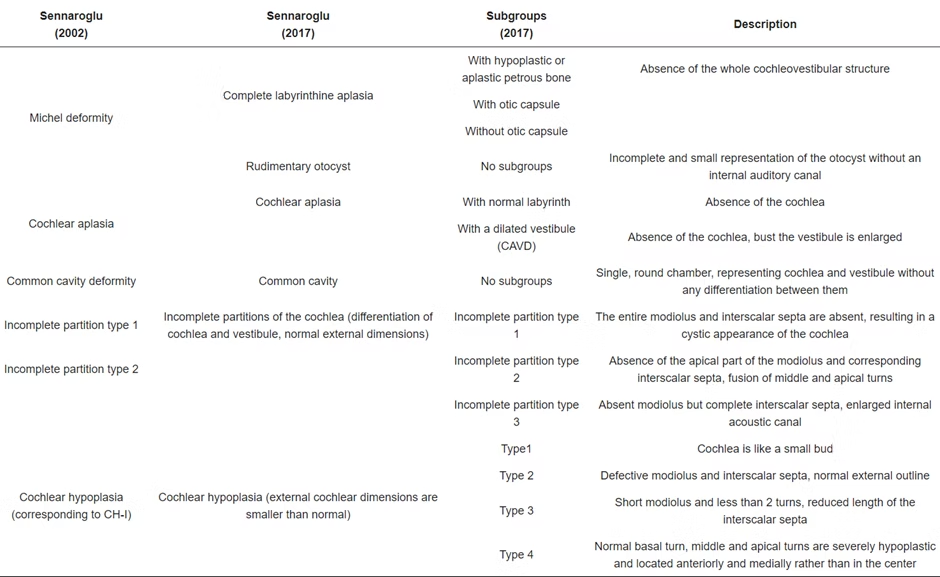
Early identification of hard of hearing since birth becomes utmost important with newborn hearing screening having a major role. Early detection, workup and classification of disorder and thereafter planning for appropriate rehabilitation is necessary.
In the landmark article that talks about the latest classification 2017 (1), the authors propose the possible imaging findings to look for by clinicians, Facial nerve anomalous routes that may coexist and the possible treatment of such malformations in order to rehabilitate hearing (HA: Hearing aid, ABI: Auditory brainstem Implant, CI: cochlear implant) (1,4,5)
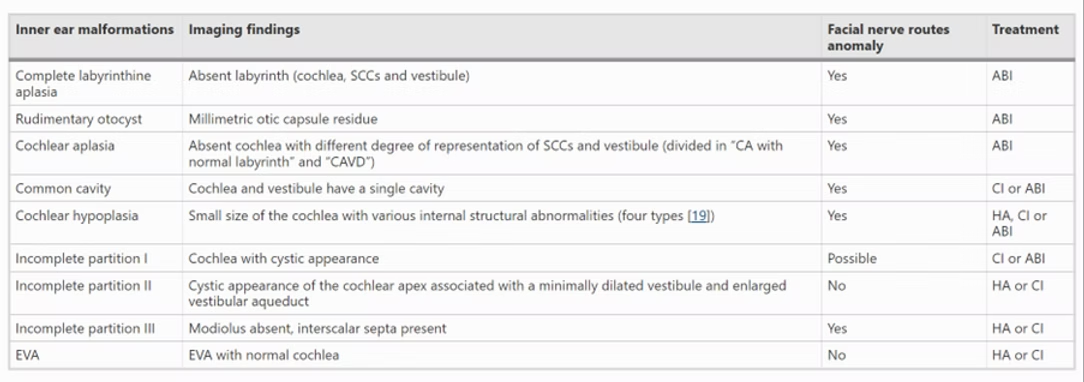
Of significance are the following considerations:
1. Classification of Inner ear malformation
2. Status of the cochlear nerve
3. Preoperative audiological findings.
The following tabular columns give the Indication of ABI and characteristic based management of inner ear malformations (1)

Management of Inner ear malformations
Predominant challenges in management are:
1.Cerebrospinal fluid gusher and risk for meningitis
2. Facial nerve anomalies
3. Decision making for the surgical approach and the type of electrode
4. Choosing the correct implantation method; CI vs ABI
5. Timing of surgery
Therefore, thorough preoperative assessment and counselling of the patient along with appropriate decision making based on universal guidelines becomes of utmost importance.
However, the final decision of rehabilitation in each patient must be done on a case-to-case basis on appropriate clinical evidence along with the experience of the surgeon in the accord of managing such cases.
One such case managed by our centre deserves a special mention where the child presented to Bangalore Head and Neck sciences at 2 years of age with a sensorineural deafness profile and was diagnosed with multiple anomalies being Incomplete partition II on one side and Absent cochlea on the other.
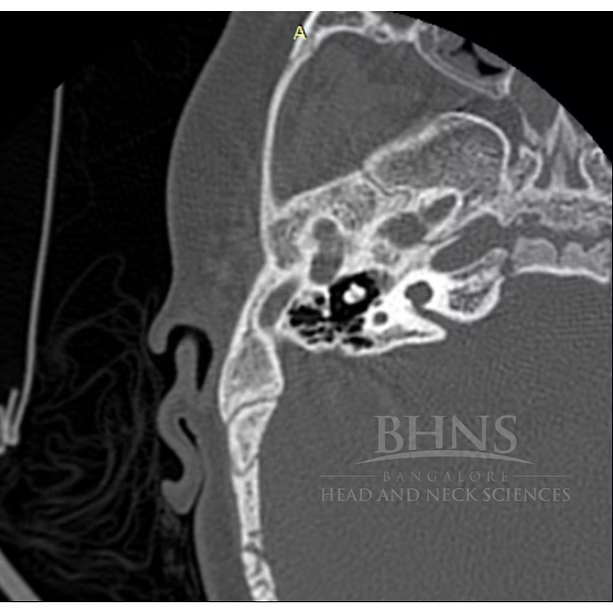
CT image 1: IPII with cystic apex of the cochlea and enlarged vestibular aqueduct

CT Image 2: Cochlear aplasia on the opposite side.
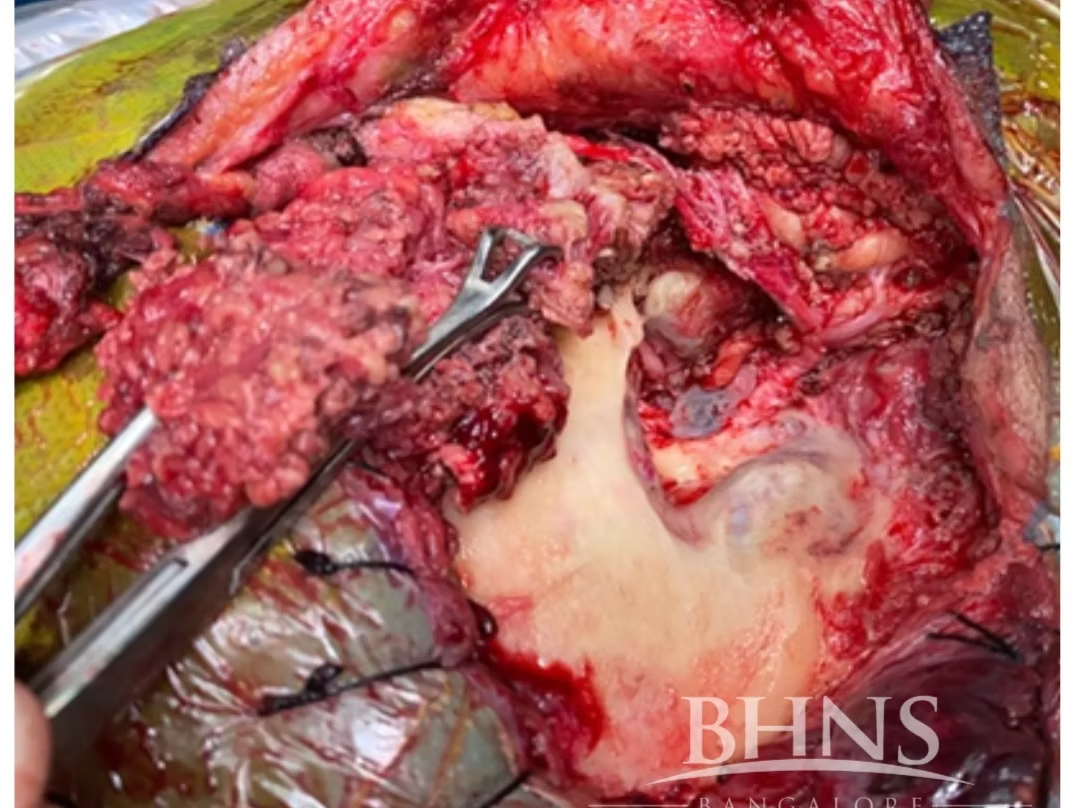
Child was told the need to undergo ABI and that the rehabilitation will be poor and challenging. At our centre the decision of CI was made. Right sided Meningoencephalic herniation into the site of the surgery was present. The course of the facial nerve was highly anomalous. The abnormal vascular malformation of the sigmoid dural venous sinus interfering with the surgical field along with middle fossa dura and posterior fossa dura left a very compact space for operating. The child underwent cochlear implantation of the right side along with Subtotal Petrosectomy.The child is now successfully able to vocalize sounds. Currently undergoing AVT sessions and is on follow up.
Hence, case-based decision making, and appropriate case selection based on thorough evidence based medicine along with expertise of the surgeon to manage complications associated with difficult anomalous Temporal bones with cochlear anomalies becomes extremely important.
Take home messages:
Early diagnosis and workup for status of hearing loss with inner ear malformation. Thorough detailed pre operative assessment and anticipation of malformations and challenges on table to be accounted for. Final decision on method of rehabilitation depends on the surgeon’s skill, clinical acumen, and the multitude of guidelines available are mere chalked out plans, and each case must be dealt with individually and successfully. Here at Bangalore Head and Neck sciences, we regularly manage disorders of the inner ear with our detailed protocols and management techniques thereby having not only multiple difficult cases that have been managed meticulously but also multiple successful rehabilitative stories gone and ongoing. For further information, visit our clinic and meet our clinicians today!
References:
1. Sennaroğlu, L.; Bajin, M.D. Classification and Current Management of Inner Ear Malformations. Balk. Med. J. 2017, 34, 397–411. 2. Brotto, D.; Uberti, A.; Manara, R. From Mondini to the latest inner ear malformations’ classifications: An historical and critical review. Hear. Balance Commun. 2019, 17, 241–248. 3. Brotto, Davide, Flavia Sorrentino, Roberta Cenedese, Irene Avato, Roberto Bovo, Patrizia Trevisi, and Renzo Manara. 2021. "Genetics of Inner Ear Malformations: A Review" Audiology Research 11, no. 4: 524-536. https://doi.org/10.3390/audiolres11040047 4. Sennaroglu L, Saatci I (2002) A new classification for cochleovestibular malformations. Laryngoscope 112:2230–2241 5. Sennaroğlu L, Tahir E (2020) A novel classification: anomalous routes of the facial nerve in relation to inner ear malformations. Laryngoscope
Temporal Bone Malignancy
Jan 18, 2023 (6 min read)Temporal bone malignancies are rare tumours accounting for less than 1 percent of all head and neck malignancies. Sun exposure is...
 Dr.
Niveditha Damodharan
Dr.
Niveditha Damodharan
Temporal Bone Malignancy
Temporal bone malignancies are rare tumours accounting for less than 1 percent of all head and neck malignancies. Sun exposure is associated with cutaneous malignancies and radiation exposure with Squamous Cell carcinoma. Chronic Otitis media has also been linked to temporal bone malignancy by some researchers. The most common malignancy seen is Squamous Cell carcinoma and other malignancies have been sparsely reported. Because of the rarity of these tumours, temporal bone is more likely to be involved in secondary malignancies arising from Parotid region or Periauricular skin cancers.
Origin and Spread
Most temporal bone neoplasms are epithelial neoplasms arising from the middle and inner ear including Squamous Cell Carcinoma, Endolymphatic sac tumor and Adenoid cystic carcinoma. Secondary tumours include those arising from Parotid gland, Nasopharynx, Brain and Periauricular Skin cancers. Temporal bone malignancies can arise from any part of the temporal bone including the External auditory canal, middle ear, mastoid, endolymphatic sac, petrous apex and the internal auditory canal. They can be locally aggressive due to the presence of numerous bony pathways in the temporal bone along which the tumor can spread from the site of origin.
Symptomatology
Ear discharge, Hearing loss and Ear ache form the classical triad of symptoms of Temporal bone malignancies. Long standing ear discharge, particularly resistant to conservative treatment should arise the suspicion of malignancy. Discharge can be purulent or bloody. Hearing loss can be purely conductive if the disease is confined to middle ear and ear canal. Sensorineural hearing loss occurs due to inner ear involvement. Tinnitus, vertigo and imbalance can also occur in such cases. Headache signals extension of the tumour intracranially. Trismus (Anterior involvement into Temporomandibular joint), Facial weakness, Involvement of other cranial nerves, Neck node metastasis will mean that the disease is advanced. Adenoid cystic carcinoma can present as masses beneath the skin of the external auditory canal.
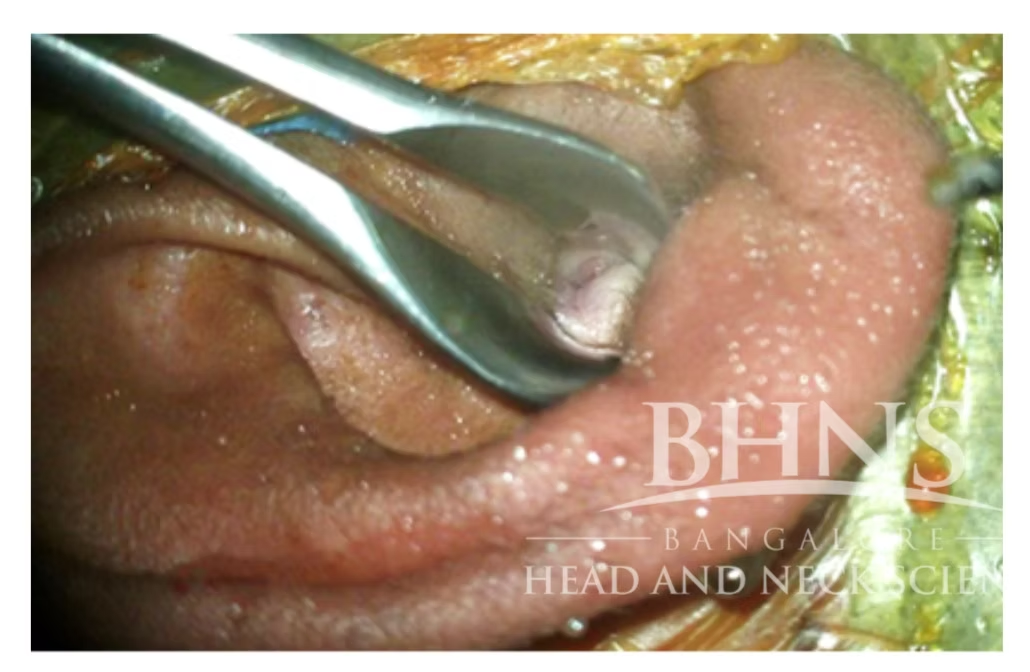
Figure 1 showing mass in the external auditory canal on examination
Diagnostic Imaging
High Resolution Computed Tomography and Magnetic Resonance Imaging with contrast are required to evaluate the extent of the tumour. CT imaging is the best to demarcate bone involvement. MRI is superior to CT scan to delineate soft tissue involvement, perineural spread, involvement of Sigmoid sinus, Internal Carotid Artery and intracranial extension. Usually anterior and inferior spread of tumour are accurately assessed radiologically, compared to superior, medial and posterior extensions1.
Staging
No proper TNM staging system has been approved by the American Joint Committee on Cancer for temporal bone malignancy. Modified Pittsburgh system was initially proposed for Squamous Cell Carcinoma of the external auditory canal and doesn’t take into account the other subsites of the temporal bone. It also doesn’t include non-squamous malignancies. However it is the most commonly used staging system. Early-stage T1 tumors are limited to the ear canal without any bony erosion or soft tissue involvement. T2 tumors have limited bony ear canal erosion or limited soft tissue involvement. T3 tumors erode the bony ear canal or have limited soft tissue involvement or begin to involve the middle ear or mastoid. T4 are large tumors that involve the inner ear, the carotid canal, the jugular foramen, and the dura or have evidence of facial paresis. Neck nodal metastasis immediately progresses the tumour stage to stage IV.
Management
Surgery remains the standard of care for Temporal bone malignancies. Adjuvant Radiotherapy has been advised for T2 and larger tumours. Positive margins, extracapsular invasion, nodal metastasis, bony or perineural invasion are other indications for adjuvant therapy. Chemotherapy has also become an emerging adjuvant option for advanced malignancies2.
Surgical management can range from Sleeve Resection to Lateral temporal bone resection (LTBR) to Subtemporal bone resection (STBR) to Total temporal bone resection (TTBR). We do not recommend Sleeve Resection as it is difficult to achieve adequate margin. Lateral Temporal bone resection is the procedure of choice for T1 and T2 tumours with or without Superficial Parotidectomy. Both LTBRs and STBRs are accepted modalities of treatment for T3 lesions. Although TTBRs are proposed as the best modality by other centres, we prefer to do an STBR with a combination of en-bloc and piecemeal resections followed by Adjuvant Radiotherapy. Due to the increased morbidity and no proven survival benefit, we do not recommend TTBR over STBR in T4 tumours.
1. Lateral Temporal Bone Resection
This procedure involves resection of temporal bone lateral to Facial nerve and is the real workhorse of Otologic Oncologic surgeries. The procedure starts with a canal wall up mastoidectomy with extended Facial recess opening. The EAC is resected en bloc along with the tympanic membrane, the malleus after disarticulation and removal of the incus, with the medial limit defined at the level of the incudostapedial joint. Superficial parotidectomy can be done with lateral temporal bone resection (LTBR), specially in T2 tumours.
2. Subtemporal Bone Resection
This procedure is an extension of LTBR and proceeds with piecemeal removal of the tumour after the initial steps of LTBR are performed. It involves IAC identification, facial nerve exposure and removal of the otic capsule with preservation of the petrous apex. Capsule of Temporomandibular joint and condyle of mandible are removed in case of anterior extension. Dural extension may warrant middle and posterior fossa craniotomies. Facial nerve is re-routed unless otherwise involved by tumour and need to be sacrificed. Sigmoid sinus and Jugular bulb can be preserved unless infiltrated. In case of doubtful tumour clearance, a vascular clip can be left behind so that postoperative radiotherapy can be targeted at that site.
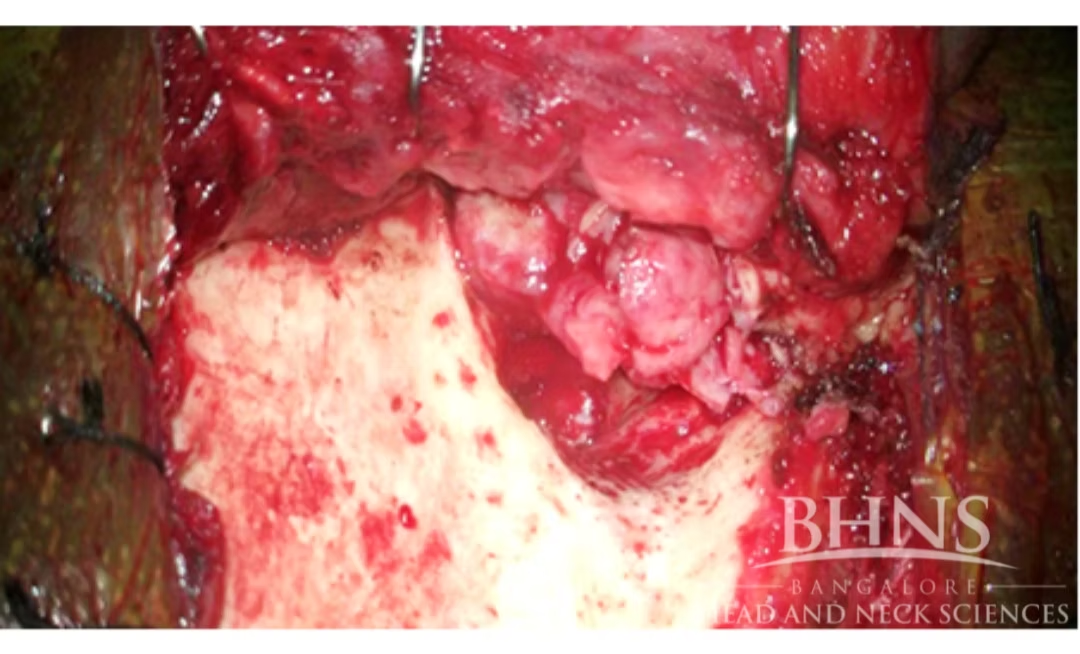
Figure 2 showing the mass in the anterior wall of EAC after elevation of flaps

Figure 3 showing tumour removal in the same patient following a Subtemporal Bone Resection with Subtotal Parotidectomy and Temporalis muscle Resection
3. Total Temporal Bone Resection
This procedure is done in advanced T4 tumours. Pinna may or may not be resected. Bone is resected superiorly for 3 cm above the temporal line to expose the middle fossa dura and behind the sigmoid sinus by a similar amount to leave a residual margin of healthy bone. Medial dissection extends through the labyrinth and exposes the intrapetrous carotid artery. Inferiorly, the sigmoid sinus and jugular bulb are mobilized from surrounding bone. The sternocleidomastoid and digastric muscles are freed from the mastoid tip. The ascending ramus of the mandible is transected and removed along with the head and coronoid process. A total parotidectomy is done and the specimen is removed en bloc. The residual tip of the petrous bone is then removed with a high-speed drill. Resection of the carotid artery can also be accomplished if the contralateral cerebral blood supply has been proven to be adequate by angiography and preoperative balloon occlusion. Overall this procedure is highly morbid and has no proven survival benefit over STBR.
Piecemeal resection is preferred over en bloc resection in large tumours to minimize morbidity and preserve vital structures3. Bone is drilled until healthy bone appears.
Role of Parotidectomy and Neck Dissection
Parotid region can be involved by either direct extension through Foramen of Huschke and Fissures of Santorini or nodal dissemination to the intra-parotid or peri-parotid lymph nodes. We do not routinely perform Superficial Parotidectomy for all T2 tumours and prefer to do it in T2 tumours with involvement of anterior wall of EAC. In T3 tumours, we routinely include Superficial Parotidectomy. In T4 tumours, a total Parotidectomy is performed.
Lymphatic drainage of EAC and middle ear goes to intra-parotid and peri-parotid, pre and postauricular, submandibular, upper deep cervical and the retropharyngeal lymph nodes. Nodal metastasis has a poor prognosis. We recommend a routine neck dissection of parotid and level 2 nodes for clinically positive nodes. For N0 necks, a frozen section of level 2 nodes can be done and can proceed to neck dissection if positive for metastasis.
Hearing Rehabilitation
Maximal conductive hearing loss occurs with LTBR and can be rehabilitated with Bone anchored hearing implants (BAHA / OSIA). Single sided deafness from STBR and TTBR can be rehabilitated with Bone anchored hearing implants or CROS hearing aids Contralateral routing of signals).
Conclusion
With the advancement in neuroimaging, neuroanesthesia and skull base surgery, temporal bone malignancies can be treated surgically with minimum morbidity. However definite protocols can be formed only after proper staging systems are put into place. Further research into the different histological varieties is also required.
Bibliography
1. Leonetti JP, Smith PG, Kletzker GR, Izquierdo R. Invasion patterns of
advanced temporal bone malignancies. Am J Otol 1996; 17:438–442.
2. Gidley PW, DeMonte F. Temporal bone malignancies. Neurosurgery
Clinics.
2013 Jan 1;24(1):97-110.
3. Prasad SC, D’Orazio F, Medina M, Bacciu A, Sanna M. State of the art
in
temporal bone malignancies. Current Opinion in Otolaryngology & Head and
Neck Surgery. 2014 Apr 1;22(2):154-65.
FACIAL NERVE SCHWANNOMA – SYMPTOMATOLOGY & MANAGEMENT
Dec 23, 2022 (5 min read)Facial Nerve Schwannomas are rare tumors arising from the Facial Nerve. They arise from the Schwann cells, which form the protective...
 Dr.
Niveditha Damodharan
Dr.
Niveditha Damodharan
FACIAL NERVE SCHWANNOMA – SYMPTOMATOLOGY & MANAGEMENT
Facial Nerve Schwannomas are rare tumors arising from the Facial Nerve. They arise from the Schwann cells, which form the protective sheath around nerves. These slow growing benign tumors occur anywhere along the course of the Facial Nerve from Cerebellopontine angle (CPA) to its extratemporal course in the Parotid gland Facial Nerve Schwannomas are rare tumors arising from the Facial Nerve. They arise from the Schwann cells, which form the protective sheath around nerves. These slow growing benign tumors occur anywhere along the course of the Facial Nerve from Cerebellopontine angle (CPA) to its extratemporal course in the Parotid gland
ANATOMY
Facial nerve is a mixed Cranial nerve consisting of 2 roots- Motor root and Nervus Intermedius. Motor root arises from Facial Nucleus at the level of Pons. Part of Facial nerve which arises from the Sensory root is called Nervus Intermedius or Nerve of Wrisberg. Special Sensory taste fibres arise from Nucleus Tractus Solitarius whereas Parasympathetic secretomotor fibres arise from Superior Salivatory Nucleus. The two roots form the Facial Nerve after they enter the anterosuperior quadrant of Internal Acoustic Meatus (IAM). The next Labyrinthine segment is the narrowest and shortest and ends in the Geniculate Ganglion which gives rise to the first branch, Greater Superficial Petrosal Nerve. The nerve runs anteromedially on the surface of the temporal bone, enters the Vidian canal and carries secretomotor fibres to the lacrimal glands. The Geniculate Ganglion contains cell bodies of the sensory neurons in the Facial Nerve. The Facial Nerve takes a 75° anteroposterior turn and becomes the Tympanic segment which then runs inside the Fallopian Canal. The Nerve makes a second turn downwards at the Pyramidal Eminence to form the mastoid segment. Nerve to Stapedius and Chorda Tympani are the other intratemporal branches. The Nerve then exits at the Stylomastoid Foramen and gives rise to two muscular branches to Posterior belly of Digastric and Posterior auricular muscles. The Nerve enters the Parotid gland where it divides into 5 distal branches –Temporal, Zygomatic, Buccal, Marginal Mandibular and Cervical.
Understanding the anatomy of the Facial Nerve is important to diagnose tumors arising from the Facial Nerve. Facial Nerve Schwannomas usually involve multiple segments of the Facial Nerve. Skip lesions can also occur. The Perigeniculate area is the commonest site of involvement followed by tympanic and mastoid segments1.
SYMPTOMATOLOGY
Symptoms are dependent on the location of the tumor. Patients usually present with Facial Paralysis2. Progressive Facial Paralysis with Facial twitching is a classical symptom of this condition. Some patients may get misdiagnosed as Bell’s Palsy. Patients can remain completely asymptomatic until the tumor becomes large enough to cause clinically observable Facial dysfunction. Perigeniculate and Intracanalicular tumors can lead to Sensorineural hearing loss. These patients can also present with tinnitus and vertigo. Lesions arising from tympanic and mastoid segment can present as middle ear masses with Conductive hearing loss. Extra-temporal involvement results in a mass in the Parotid region. A tumor involving the Greater Superficial Petrosal Nerve can present as an extra-axial mass in the Middle Cranial Fossa along with dry eye. Dysgeusia can occur if the nerve is involved near the origin of Chorda Tympani.
RADIOLOGY
High Resolution Computed Tomography of the Temporal bone would show smooth osseous widening of the Fallopian Canal. Schwannomas usually appear iso- to hypointense relative to gray matter on T1-weighted images and hyperintense on T2 weighted images. T1 Contrast Magnetic Resonance Imaging would usually show FNS as well-circumscribed fusiform enhancing masses along the course of the Intratemporal Facial Nerve. Enhancement in MRI with widening of the Fallopian Canal is diagnostic of FNS3. Larger tumors may undergo cystic degeneration seen as focal intramural low signal intensity on contrast-enhanced T1 images4. FNS involving the CPA-IAM facial nerve segments cannot be distinguished from Vestibular Schwannomas if extension into the labyrinthine segment of the facial nerve is not present. The presence of a Labyrinthine tail is indicative of a FNS. Extensive FNS involving CPA-IAM and Perigeniculate area do not conform to the usual radiological presentation and cause dumb bell shaped masses on imaging4. Tympanic segment FNSs are often multi-lobular and they extend into the middle ear to present as an avascular retrotympanic mass with conductive hearing loss. Finally, mastoid segment FNSs break into the surrounding thin wall septations of the mastoid air cells and can seem as locally aggressive masses on MR imaging.
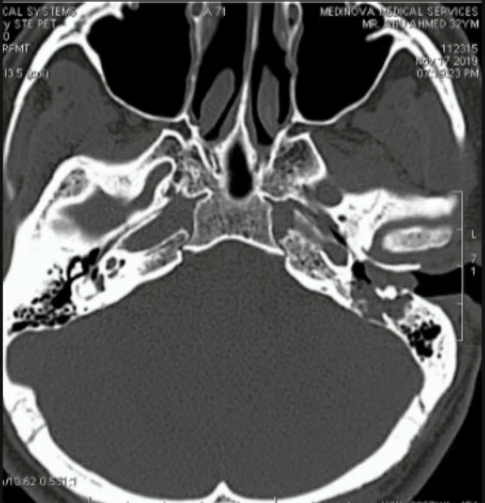
Figure 1 showing Facial Nerve Schwannoma presenting as a aural polyp
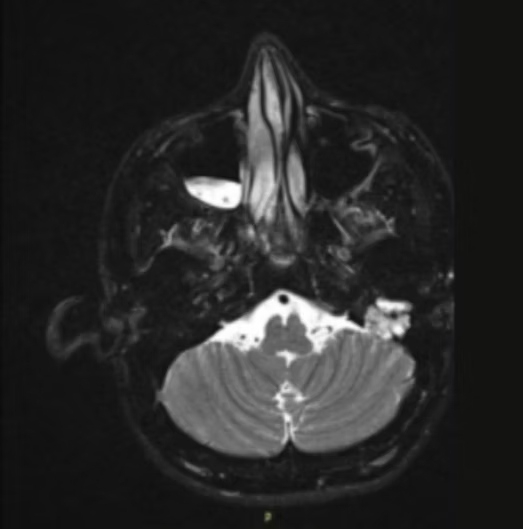
Figure 2 MRI scan shows Hyperintense mass arising from Facial Nerve on the left side.
MANAGEMENT
Surgery remains the mainstay of treatment. Surgical decision making depends on the site, size of the tumour , preoperative hearing and Facial nerve status. Some form of Facial paresis is expected postoperatively. Hence an informed decision should be taken in such cases. In cases where there is a slow growing tumour ( 1.4mm /year)5 with preserved Facial nerve function, watchful waiting would be the best option until Facial nerve function worsens to House Brackmann ( HB ) grade III. In cases with HB> III Facial nerve dysfunction, Surgery would be preferred.
Management
Surgery remains the mainstay of treatment. Surgical decision making depends on the site, size of the tumour , preoperative hearing and Facial nerve status. Some form of Facial paresis is expected postoperatively. Hence an informed decision should be taken in such cases. In cases where there is a slow growing tumour (1.4mm /year)5 with preserved Facial nerve function, watchful waiting would be the best option until Facial nerve function worsens to House Brackmann ( HB ) grade III. In cases with HB> III Facial nerve dysfunction, Surgery would be preferred.
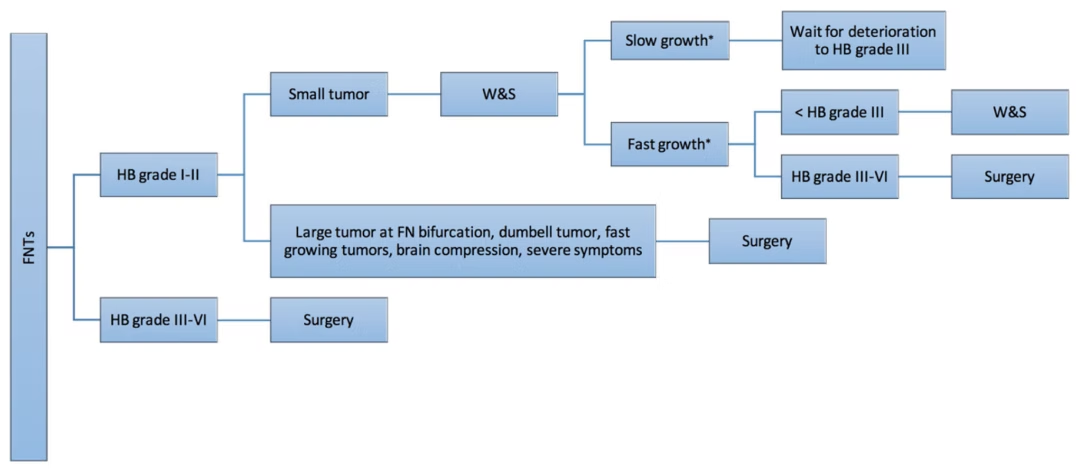
Figure 3 explains the decision making algorithm proposed by Sampath3 et al.
Surgical management can range from Sleeve Resection to Lateral temporal bone resection (LTBR) to Subtemporal bone resection (STBR) to Total temporal bone resection (TTBR). We do not recommend Sleeve Resection as it is difficult to achieve adequate margin. Lateral Temporal bone resection is the procedure of choice for T1 and T2 tumours with or without Superficial Parotidectomy. Both LTBRs and STBRs are accepted modalities of treatment for T3 lesions. Although TTBRs are proposed as the best modality by other centres, we prefer to do an STBR with a combination of en-bloc and piecemeal resections followed by Adjuvant Radiotherapy. Due to the increased morbidity and no proven survival benefit, we do not recommend TTBR over STBR in T4 tumours.
Surgical approach would depend on Hearing status. A Middle Cranial Fossa approach would be suitable for a tumor proximal to the geniculate ganglion with serviceable hearing, provided the tumor does not extend far into the cerebellopontine angle. A Retrosigmoid approach gives the best chance of hearing conservation in CPA lesions >1 cm. With non-serviceable hearing, a translabyrinthine approach is the procedure of choice. This approach also provides the best access for Facial Nerve grafting.

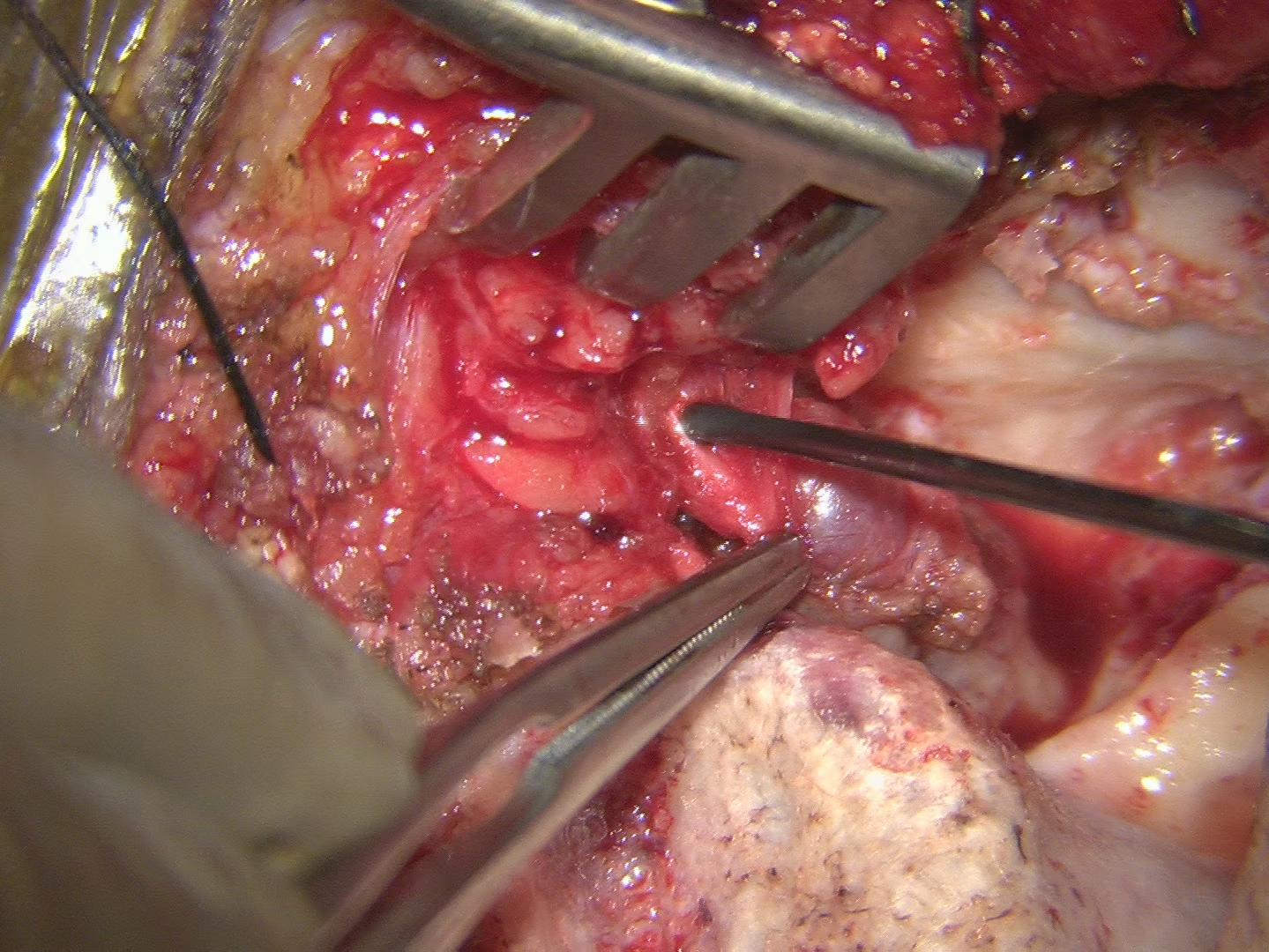
Figure 4 showing an intraoperative picture of Facial Nerve Schwannoma.
Management of Facial nerve can range from preservation of nerve integrity to grafting to anastomosis3. Good Facial nerve function cannot be guaranteed even in cases where the surgeon tries to preserve the integrity of the nerve. Primary anastomosis even with nerve re-routing may compromise neural blood supply. Interposition grafting is usually performed with Greater Auricular or Sural nerves, but this doesn’t give favourable results if the Facial nerve paresis has been present > 1 year. A XII to VII or a V to VII anastomosis is preferred in such cases. If this fails, dynamic facial reanimation techniques such as Labbe’s surgery is used. It has to be noted that full facial recovery isn’t possible in all these procedures and the maximum function achieved ranges from HB III-1V.
CONCLUSION
Management should be tailored according to tumor location and size, existing facial nerve function, patient priorities and age, with priority placed on function preservation over complete tumor removal.
BIBLIOGRAPHY
1. Wilkinson EP, Hoa M, Slattery III WH, Fayad JN, Friedman RA, Schwartz
MS,
Brackmann DE. Evolution in the management of facial nerve schwannoma.
The
Laryngoscope. 2011 Oct;121(10):2065-74.
2. Chung JW, Ahn JH, Kim JH, Nam SY, Kim CJ, Lee KS. Facial nerve
schwannomas: different manifestations and outcomes. Surgical neurology.
2004
Sep 1;62(3):245-52.
3. Prasad SC, Laus M, Dandinarasaiah M, Piccirillo E, Russo A, Taibah A,
Sanna M. Surgical management of intrinsic tumors of the facial nerve.
Neurosurgery. 2018 Oct 1;83(4):740-52.
4. Wiggins RH, Harnsberger HR, Salzman KL, Shelton C, Kertesz TR,
Glastonbury CM. The many faces of facial nerve schwannoma. American
journal
of neuroradiology. 2006 Mar 1;27(3):694-9.
5. Perez R, Chen JM, Nedzelski JM. Intratemporal facial nerve
schwannoma: a
management dilemma. Otol Neurotol 2005; 26(1):121-126.
PETROUS BONE CHOLESTEATOMA
Nov 18, 2022 (3 min read)Cholesteatoma is an abnormal collection of skin deep inside the ear. Petrous bone cholesteatoma (PBC) refers to an epidermoid cyst...
 Dr.
Niveditha Damodharan
Dr.
Niveditha Damodharan
PETROUS BONE CHOLESTEATOMA
Cholesteatoma is an abnormal collection of skin deep inside the ear. Petrous bone cholesteatoma (PBC) refers to an epidermoid cyst occurring in the petrous portion of the temporal bone. It is an unusual entity occurring in about 4-6% of all temporal bone lesions1. It is slow growing and hence can remain undetected until it involves the facial nerve or labyrinth.
Why does a Petrous bone Cholesteatoma occur?
PBCs can be Congenital, Acquired, or Iatrogenic2. Congenital PBC occurs due to the epidermal cells resting within the temporal bone and presenting as a pearly white mass behind an intact tympanic membrane. A diagnosis of congenital PBC is considered when there is no previous history of otorrhea, trauma, or surgery. A large primary acquired cholesteatoma occurring in a well-pneumatized temporal bone can extend above or below the labyrinthine compartment to form a PBC. The iatrogenic type happens due to the implantation of squamous epithelium after ear surgery.
Clinical Presentation of a PBC
PBCs are locally destructive lesions yet patients remain asymptomatic until the disease involves vital structures. The commonest presenting symptom is progressive conductive or sensorineural hearing loss. Progressive or sudden facial weakness can cause alarm. Patients can also present with vertigo, tinnitus, imbalance, foul smelling ear discharge. Headache, vomiting, and seizures occur in case of intracranial extension/complications. Involvement of other cranial nerves can cause numbness of the face, Double vision, and Trigeminal neuralgia.
How do we diagnose a PBC?
Diagnosis would involve a complete neurotological examination and imaging. A high-resolution computed tomography of the temporal bone with 1mm cuts will help delineate the extent of the lesion and aid in surgical planning. With the advent of Magnetic Resonance imaging, it has become increasingly easier to distinguish PBCs from other temporal bone lesions. Diffusion Weighted images are particularly useful in identifying residual/recurrent PBCs. Audiometric tests are performed to assess the type and degree of hearing loss. They are also needed for planning hearing rehabilitation after surgery.
Figure 1 shows a high resolution computed tomography scan of the temporal bone with a left Petrous bone cholesteatoma.
How to treat?
Surgery remains the mainstay of treatment for Petrous bone Cholesteatoma. The main goal of surgery is the full eradication of disease and to give a dry, trouble-free ear. Advances in Neuroradiology and neuromonitoring have made it possible to achieve good results with minimum morbidity. The surgical approach depends on the site and type of the lesion, degree of hearing loss, and integrity of the facial nerve. Sanna et al.,3 have classified PBCs into 5 types namely Supralabyrinthine, Infralabyrinthine, Infralabyrinthine-apical, Massive and Apical. Subclasses were added by Sampath et al.,2 vis-à-vis extension to clivus, sphenoid sinus, nasopharynx, and intradural. Commonly used approaches are Transotic ( preferred ), Modified transcochlear Type A and B, Subtotal Petrosectomy, Translabyrinthine, and Infratemporal fossa type B approach.

Figure 2 shows an intra-operative picture of a left sided Petrous bone Cholesteatoma
Management of the Facial nerve
Transotic approach is preferred in cases of preoperative preserved facial nerve function. Some form of facial nerve management would be essential in all cases and can range from skeletalization or decompression of the nerve to Active management. The latter can involve
1. Partial / Total Posterior re-routing of the nerve
2. Posterior re-routing and end-end anastomosis
3. Nerve sectioning and end-end anastomosis
4. Nerve sectioning and cable nerve grafting (Sural nerve is used)
5. Facial- Hypoglossal or Facial- Trigeminal anastomosis.
Hearing Rehabilitation
Hearing preservation surgery can be performed in limited Supralabyrinthine (Transmastoid - Middle cranial fossa approach) or Infralabyrinthine (Transmastoid- Retrofacial approach / Subtotal Petrosectomy) PBCs.
However, in the presence of a fistula in the cochlea, the Middle cranial fossa approach cannot preserve hearing.
Generally, complete disease removal and preservation of facial nerve function take precedence over hearing preservation. Hearing can be rehabilitated in such cases with bone-anchored hearing aids or cochlear implants or Soundbridge in the same sitting.
PBCs are surgically challenging and management requires meticulous preoperative evaluation, planning, execution, and strict follow-up. Radical surgery to eradicate the disease and preserve facial nerve function remains a cornerstone of management.
References
1. Omran A, De Donato G, Piccirillo E, Leone O, Sanna M: Petrous bone
cholesteatoma: management and outcomes. Laryngoscope 2006;116: 619–626.
2. Prasad SC, Piras G, Piccirillo E, Taibah A, Russo A, He J, Sanna M.
Surgical strategy and facial nerve outcomes in petrous bone
cholesteatoma.
Audiology and Neurotology. 2016;21(5):275-85.
3. Sanna M, Zini C, Gamoletti R, et al. Petrous bone cholesteatoma.
Skull
Base Surg 1993;3:201-13.
Radiology and Treatment of Skull Base Osteomyelitis
Oct 26, 2022 (7 min read)Radiology and treatment of SBO Skull base osteomyelitis (SBO) is a debilitating progressive disorder occurring in immunocompromised...
 Dr.
Ajay Bhandarkar
Dr.
Ajay Bhandarkar
Radiology and Treatment of Skull Base Osteomyelitis
Radiology and treatment of SBO
Skull base osteomyelitis (SBO) is a debilitating progressive disorder occurring in immunocompromised individuals. It commonly affects the bony external auditory canal with extension to involve the neighbouring bony and soft tissue structures. In this blog, we aim to discuss the radiology and treatment aspects of Skull base osteomyelitis.
Radiology
Skull base Osteomyelitis(SBO) presents with diverse manifestations with involvement of bone and soft tissue. Extension into the intracranial compartment producing debilitating complications in uncontrolled SBO is not uncommon. Various inflammatory pathologies and malignancies mimic SBO, hence, it is paramount that an appropriate radiological investigation is chosen in the diagnosis and prognosis of SBO.
High-Resolution CT Imaging
The primary purpose of doing a high-resolution CT scan with contrast administration is to determine the erosion of bone. Subtle cortical erosions, thickening of external auditory canal skin, and fat plane effacement in the stylomastoid foramen and infratemporal fossa regions are the early manifestations, however, these findings are not specific to SBO. CT scan can pick up bony erosion only if there is a minimum of 30% bony demineralization due to osteolysis caused by the underlying pathology. Hence, a CT scan even with its exceptional anatomic resolution exhibits poor sensitivity and specificity in early diagnosis of SBO, however, the sensitivity improves when there is a locally advanced SBO i.e SBO. The advantage of a CT scan is that it is widely available and inexpensive when compared to other radiological investigations used in the diagnosis of SBO.
MRI With Gadolinium Contrast
Soft tissue assessment is superior to MRI when compared to a CT scan. T1-weighted images demonstrate a hypointense signal in the external auditory canal and subtemporal region soft tissues and an isointense to minimal hyperintense signal on T2-weighted images. Most of the inflammatory conditions exhibit hyperintense signals on T2-weighted MRI due to hyperemia and edema, unlike SBO which exhibits isointense or mild hyperintense signals due to the compromised vascular supply primarily due to diabetic microangiopathy and necrotizing pathology. Gadolinium contrast administration may demonstrate diffuse or focal rim-enhancing fluid collection due to inflammation. Fat suppression following contrast administration accurately determines skull base enhancement. Dural enhancement and bony medullary space involvement is also a feature of MRI which makes it superior to CT scan. The advantage of MRI is there is no radiation burden to the patient. MRI is also superior to CT imaging in detecting the anatomical site of involvement. Diffusion-weighted imaging(DWI) on MRI has an additional advantage in bacterial SBO wherein, an enhanced apparent diffusion coefficient helps it to differentiate from lymphoma and malignancy.
Both MRI and CT are poor in therapeutic prognostication of SBO as the bone and soft tissue changes remain for a period of 6 months to 1 year after the disease has resolved.
Nuclear Imaging
Beta-emitting tracers and gamma tracers have been used in SBO to aid in early diagnosis and follow-up. These tracers form an important component of scintigraphy, SPECT, and PET. The inherent disadvantage of this imaging modality is the poor anatomical detail and spatial resolution.
(a) Gamma tracers
Technitium 99m-methylene diphosphonate detects osteoblastic activity. Any condition with an increased bone turnover is detected by this radiotracer. It detects even a 10% enhancement of activity, thereby making it beneficial in the early stages of SBO. It cannot be used effectively in the detection of treatment response as osteoblastic activity persists post-therapy. Technitium-labeled leukocytes detect infectious foci. However, the disadvantage of using it is the expense involved and its inability to detect low-grade infections. Gallium-67-citrate binds to vigorously dividing leukocytes in SBO. It is extremely useful in detecting the therapeutic response. However, it delivers a high radiation dose and is expensive.
(b) Beta tracers
FDG (2-Fluoro-2-desoxy-glucose) detects enhanced metabolism and is not a specific infection tracer as it is highly sensitive and detects any pathology with increased metabolic activity.
Meta-analysis of nuclear imaging in SBO revealed a sensitivity of 82%, 61%, 78%, 84% and specificity of 25%, 77%, 84%, and 60% for bone scintigraphy, leukocyte scintigraphy, combined bone-leukocyte scintigraphy, and MRI respectively. 18F-FDG PET/CT was considered a reliable diagnostic and prognostic imaging indicator until the advent of hybrid imaging. A gallium-67-citrate scan is a good indicator for the resolution of SBO. Recently 99m Tc‐HMPAO‐leukocyte scintigraphy has been found to be promising in the assessment of healing with a sensitivity and specificity of 86% and 75% respectively.
Hybrid Imaging
Hybrid imaging has emerged as the workhorse in the combined anatomical and functional detection of SBO. Technitium 99m-MDP SPECT/CT scan has reported a sensitivity of 100% and a specificity of 78%. This imaging modality can detect bony changes 24-48 hours after disease onset. FDG-PET/CT has reported a sensitivity and specificity of 96% and 91% making it a reliable imaging technique in the early detection of SBO.
Treatment
Antibacterial Therapy
Multiple studies have debated the role of monotherapy vs combination antibacterial therapy in the treatment of SBO. Resistance of Pseudomonas aeruginosa to ciprofloxacin, however, has warranted the role of combination therapy in SBO. Second, there is increasing evidence of polymicrobial infection in SBO which include S.aureus, MRSA, and anaerobic bacteria. In addition, Aspergillus, Candida, and Mycobacteria have been evident in immunocompromised patients in the 20th century.
6-week-course of culture-directed antibacterial combination therapy is the norm for SBO/SBOM which includes the 4 weeks required for bone revascularisation. Fluoroquinolone(Ciprofloxacin) and a 3rd generation cephalosporin(Ceftazidime) is the first line of treatment for SBO. Piperacillin/Tazobactam as monotherapy has shown an equally good therapeutic response. The role of additional antibiotics is limited to the presence of MRSA and anaerobic bacterial culture which may include vancomycin or metronidazole respectively. The role of gentamicin as long-term therapy is limited because of its toxicity in spite of its capability of augmenting the action of penicillin agents although it has been recommended with a prescription limited to one week. Individuals who are sensitive to the penicillin group can be safely substituted with 3rd generation cephalosporins as there is evidence of only 10% cross-sensitivity between the groups. Topical fluoroquinolones in SBO is controversial and avoided due to its ability to change the ph drastically in an already altered environment in the external auditory canal. Burow’s solution(ph 3.2) in 1:4 dilution is beneficial due to its antibacterial nature (acidic environment + aluminium acetate action). 1:4 dilution ascertains minimal discomfort in an acutely inflamed environment. Re-assessment of therapy is done by imaging at 6 weeks with repeat culture to ascertain the future course of treatment.
Avoidance of Ciprofloxacin Monotherapy as First-Line Therapy
Drug resistance is the most important cause for monotherapy avoidance. Mutation of topoisomerase IV and DNA gyrase enzymes which are primarily responsible for quinolone activity has been the dominant cause of ciprofloxacin-resistant pseudomonas. Other reasons for quinolone resistance are the prescription of improper oral and topical drug regimes for the treatment of systemic/otological conditions and the development of pseudomonas biofilms. There is also evidence of enhanced risk of C.difficile infection in an already immunocompromised patient which may have drastic consequences in long-term monotherapy.
Antifungal Therapy
Increased evidence of invasive Aspergillus species has been reported with SBO in immunocompromised individuals. Voriconazole has been proposed as the first-line antifungal therapy in the treatment of Aspergillus SBO. It has demonstrated satisfactory bone, tissue, and blood-brain barrier penetrability and superior innate anti-Aspergillus action when administered orally or intravenously. It has a better safety profile and efficacy compared to Amphotericin B(conventional or liposomal). However, it has potential hepatic adverse effects and drug interactions that mandate therapeutic drug monitoring in immunocompromised individuals. The presence of candida in SBO warrants the use of fluconazole as therapy. Long-term use of fluconazole has gastrointestinal adverse effects. Hence, Itraconazole is a viable second-line option in Candida SBO.
Antifungal therapy should be initiated under the following circumstances (1) No response to culture-directed antibacterial therapy after 6 weeks (2) Emergence of complications (nerve palsies, features of SBOM) (3) Positive fungal culture.
Antitubercular Therapy
Role of Mycobacteria in SBO can be suspected under three circumstances: (1) No response to antibacterial or antifungal therapy after 6 months as evidenced by imaging (2) Endemic mycobacterial geographical distribution (3) Positive gen expert tissue MTB assay. Literature review on mycobacterial involvement in SBO is sparse and one case report from a mycobacterial endemic zone reported remission of SBO following intensive therapy for 2 months with isoniazid, rifampicin, pyrazinamanide and ethambutol and continuation therapy with isoniazid and rifampicin for 4 months when antibacterial and antifungal therapy had failed.
Surgical Treatment
The role of surgical therapy in SBO is minimal considering the advancement in antimicrobial therapy and there are isolated case reports in literature which suggest minimal debridement to radical surgery. Peled et al in their series have suggested specific indications for surgical intervention. They are (1) Non-response to antibiotic therapy. (2) Advanced disease (nerve palsies, TMJ involvement, infratemporal fossa or nasopharyngeal involvement) or bilateral disease (3) Isolated facial paralysis (4) Deep tissue sterile culture. The surgical debridement should be able to achieve complete removal of osteomyelitic bone.
Hyperbaric Oxygen Therapy (HBOT)
Hyperbaric oxygen therapy has been proposed as an adjunct in the treatment of MOE/SBOM. Although there is no concrete evidence in the literature for the application of HBOT in MOE/SBOM, it has shown promise in advanced stages with single/multiple cranial nerve palsies, intracranial extension, or fungal MOE/SBOM. 100% oxygen is delivered at approximately 2-2.5 absolute atmosphere(ATA) once/twice daily over one to two hours for a duration ranging from 20 to 40 days in conjunction with antibiotic/antifungal therapy as per HBOT protocol. The hypoxic state produced by inflammation and osteomyelitis is retracted by 100% oxygen administration which enhances vasoconstriction and oxygen carrying capacity of the blood. It also increases the antibacterial leukocyte activity and osteal activity with a reduction in tissue edema. Sustained administration improves angiogenesis and collagen deposition thereby promoting tissue healing. The documented side effects of HBOT are acute pulmonary edema, oxygen-toxic seizures, claustrophobia, and tympanic membrane perforation.
Conclusion
Radiology forms an integral part of the treatment of skull base osteomyelitis. Impetus lies on the extension of the disease which commonly involves the cranial nerve and causes severe debility. Surgery along with medical therapy forms the prime treatment of choice in skull base osteomyelitis.

Facial Nerve Palsy
Sep 23, 2022 (3 min read)The facial nerve is the 7th cranial nerve in the human body. It arises from 4 nuclei situated at the lower pons namely, the motor...
 Dr.
Ajay Bhandarkar
Dr.
Ajay Bhandarkar
Facial Nerve Palsy
Radiology and treatment of SBO
The facial nerve is the 7th cranial nerve in the human body. It arises from 4 nuclei situated at the lower pons namely, the motor nucleus, superior salivatory nucleus(parasympathetic), lacrimatory nucleus (parasympathetic), and nucleus of Tractus solitarius (gustatory).
The nerve is a mixed nerve containing the following components:
(1) Branchial motor (special visceral efferent)
(2) Visceral motor (general visceral efferent)
(3) Special sensory (special afferent) and General sensory (general
somatic
afferent).
The entire course of the facial nerve is presented in intracranial course passage through the temporal bone from the internal auditory meatus to the stylomastoid foramen distribution to musculature to the face & scalp.
Motor and sensory roots (nervus intermedius of wrisberg ) have no epineurium and are covered by piamater and surrounded by CSF (Hence making it tensile and resistant to the slow process of stretching and compression).
The motor root supplies the muscles of the face, scalp, auricle, buccinator, platysma, stapedius, stylohyoid and posterior belly of the digastric. The special sensory root is gustatory and arises from the anterior 2/3rd of the tongue via the chorda tympani nerve. It also carries the preganglionic parasympathetic (secretomotor) of submandibular and sublingual salivary glands, lacrimal glands, and the glands of nasal and palatine mucosa.
The facial nerve passes through the internal auditory meatus to become the meatal portion and then the labyrinthine portion before finally entering the middle ear and exiting at the stylomastoid foramen to follow a tortuous course. The branches of distribution of the facial nerve are as follows:
Within the facial canal
(1) Greater superficial petrosal nerve
(2) Nerve to stapedius
(3) Chorda tympani
At the exit from the facial canal
(1) Post.auricular
(2) Digastric
(3) Stylohyoid
Terminal branches within the parotid gland
(1) Temporal
(2) Zygomatic
(3) Buccal
(4) Marginal Mandibular
(5) Cervical
Causes of facial nerve palsy
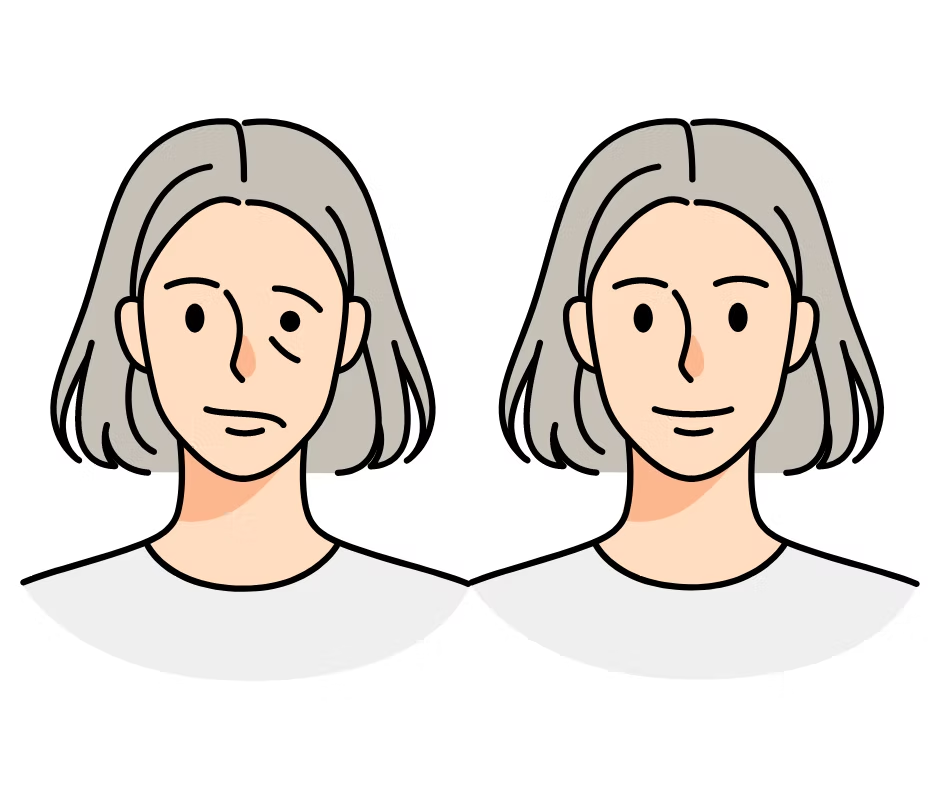
1 Congenital
Rare - due to congenital nuclear aplasia - Moebius’ syndrome
Myotonic dystrophy
Melkersson –Rosenthal syndrome
Congenital facial nerve palsy
2 Neurologic
Myasthenia gravis
Multiple sclerosis
Guillian-Barre syndrome
3 Neoplastic
Facial nerve tumors- Schwannoma, Neurofibroma, Neurogenic sarcoma
Paragangliomas
Meningiomas,
Vestibular Schwannoma
Parotid tumors
Temporal bone / external auditory canal tumors
4 Infections
Otitis media, mastoiditis
Bacterial causes (diptheria, tuberculosis)
Viral causes – herpes zoster, lyme disease ,mumps, infectious
mononucleosis
5 Iatrogenic
Mandibular block anesthesia
Parotidectomy
Lateral skull base surgery
6 Traumatic
Temporal bone fractures
Penetrating trauma (gunshot)
Facial lacerations
High altitude palsy
7 Other causes
Toxic
Metabolic
Idiopathic - Bell’s palsy, Brainstem infarction
On clinical examination, the patient might complain of facial weakness, loss of taste sensation, reduced saliva and lacrimation and reduced sensation in the external auditory canal depending on the site of involvement of the facial nerve.
Testing of facial nerve function is done by:
ELECTRO DIAGNOSIS – testing the degree of distal axonal degeneration TOPOGNOSIS – testing the function of accessory branches. ORTHODOMIC CONDUCTION - The nerve is stimulated proximally and the distal muscle response recorded ANTIDORMIC CONDUCTION – the ability of the nerve to conduct in a retrograde manner is tested
The following methods are employed to test each of the functions:
Topognosis- lacrimation, stapedial reflex, salivary flow,taste Electrodiagnosis – NET, Maximal stimulation, EMG, ENoG Intra-operative monitoring - determines the electrically evoked potential and mechanically evoked potential
Radiology plays an important role in the diagnosis of facial nerve disorders. MRI and CT scans predominantly form imaging modalities to detect facial nerve disorders with/without contrast. 3D reconstruction of CT and MRI provide a life-like display of larger structures within the temporal bone. The medical line of management depends on the etiology. A wide range of medications can be used to treat facial nerve disorders.
The indications for surgical management of facial nerve palsy are
(1) Nerve palsy progressed from incomplete to complete palsy over a
period of
hours to days
(2) Dropping response to ENoG to less than 25% of normal or dropping
steadily even after the 3rd day of the onset.
(3) Facial nerve tumor infiltration
(4) Facial nerve transection
(5) Palsy associated with CSOM (with or w/o cholesteatoma)
Surgical Modalities include
1 Decompression
(2) Direct Nerve Repair with/without using a graft
(3) Facial Reanimation: Nerve Grafting
Nerve Crossover
Muscle transposition
Free Muscle Transfer
(4) Re-routing

Squammous cell carcinoma of Temporal bone: A current review
May 17, 2022 (7 min read)Squamous cell carcinoma is the most common malignancy of the external auditory canal, middle ear, and mastoid. Surgical resection is the...
Squammous cell carcinoma of Temporal bone: A current review
Squamous cell carcinoma is the most common malignancy of the external auditory canal, middle ear, and mastoid. Surgical resection is the cornerstone of treatment, with T1 lesions of the external auditory canal treated by lateral temporal bone resection and more advanced lesions treated by subtotal or total temporal bone resection.
Clinical features
Patients with cancer of the temporal bone most often present when aged 60 years or older, although any age group, including children, can be affected.
Symptoms and signs of temporal bone lesions are summarized as follows:
· Otalgia (80-85%)
· Otorrhea (40-75%)
· Facial paralysis (25%)
· Hearing loss (45-80%)
· Tinnitus (8-10%)
· Vertigo
· Auricular lesion
· External canal mass (10%)
· Parotid mass (19%)
· Skin lesions
· CN V, IX, I, XI deficits (30%)
Workup in malignant tumors of the temporal bone
Routine preoperative testing includes complete blood counts (CBCs),
electrolyte level tests, renal function tests, liver function tests,
coagulation.
Imaging studies can include the following:
· HRCT and MRI scan- Imaging is important in TBMs for accurate tumor and node staging as many patients have limited findings on physical examination. Highresolution computed tomography of the temporal bone offers the most accurate method for the evaluation of bone erosion due to malignancies. However, a reported limitation of CT is its inability to distinguish between tumor and fluid in the middle ear, soft tissue or mucosal thickening in the absence of bone erosion. Also, spread along fascial planes and neurovascular structures can be difficult to detect. MRI can provide excellent differentiation between soft-tissue tumor margin, muscle and soft-tissue infiltration, and can help in distinguishing tumor from obstructive inflammatory changes. In addition, obstruction of the sigmoid sinus and encasement of the petrous internal carotid artery are better detected on MRI than CT, because of the vascular signal void seen on precontrast MRI and the flow enhancement of the sigmoid sinus seen on postcontrast MRI. Tumor extension, specially cranial spread into the middle and posterior cranial fossa and caudal spread into the infratemporal fossa, is also better detected on MRI. Enhanced T1-weighted spin-echo images with fat-signal suppression are most suitable for this purpose. MRI has also made it possible to confidently diagnose perineural spread of malignancies. Fat saturation gadolinium-enhanced magnetic resonance (MR) scans are often capable of detecting subtle tumor tracking along the fifth and seventh cranial nerves, as well as other nerves that travel through the many foramina of the skull base, before the lesions have grown sufficiently large to affect the surrounding bone.
· Chest radiography - If the histology indicates squamous cell carcinoma,
obtain plain radiographs or CT scans of the chest to rule out metastasis
· CT scanning of the chest, abdomen, or pelvis - This is not necessary
unless the biopsy specimen of the temporal bone tumor reveals a tumor
with a
known propensity for metastasis
· Carotid angiography with balloon occlusion Xenon test - If the carotid
artery is suspected to be involved
Other tests include the following:
· Audiometry - An audiogram is obtained prior to performing any major
procedure on the ear or temporal bone; audiograms provide baseline
hearing
thresholds for future comparison
· Biopsy - to determine whether the lesion in the ear is benign or
malignant.Histologic examination is important because, although CT
scanning
provides important preoperative staging information, systematic
pathologic
evaluation of the specimen is crucial for staging and treatment.
Management of Temporal Bone Malignancy
Surgery is the primary treatment of choice in TBMs. Radiotherapy is used as an adjuvant treatment to surgery except in advanced tumors requiring palliation. The role of chemotherapy is not established in TBMs. Surgical techniques for temporal bone malignancies
· Modified lateral temporal bone resection
· Lateral temporal bone resection
· Subtotal temporal bone resection
· Total temporal bone resection
· Lateral temporal bone resection
This is the primary surgery of choice in T1 and T2 tumors. The approach entails a complete canal wall up mastoidectomy with an extended facial recess opening. The EAC is resected en bloc along with the tympanic membrane, the malleus after disarticulation and removal of the incus, with the medial limit defined at the level of the incudostapedial joint. Some authors advocate that routine superficial parotidectomy be done with lateral temporal bone resection (LTBR), specially in T2 tumors.
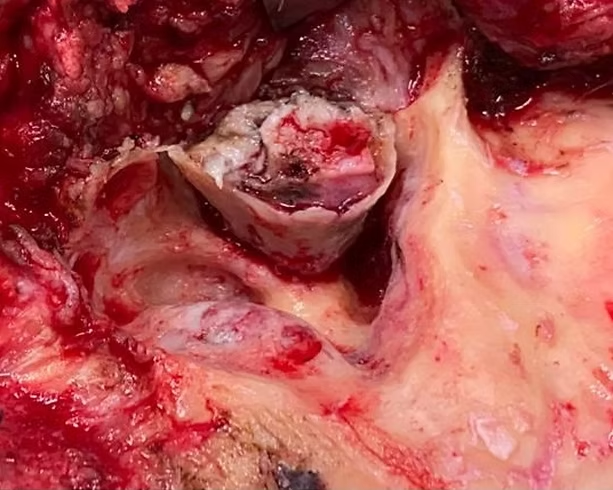
pFig2: Lateral Temporal Bone Resection
· Subtotal Temporal bone resection
This is used in T3 and T4 tumors and is an extension of the LTBR. After
the
steps of LTBR are performed, this procedure extends medially in a
piecemeal
fashion and includes IAC identification, facial nerve exposure and
removal
of
the otic capsule with preservation of the petrous apex. Care must be
taken
to
remove adequate bone around the tumor. The capsule of the
temporomandibular
joint and, if necessary, the condyle of the mandible is resected when
found
involved. If the tumor extends into the mastoid and dural involvement is
suspected, middle and posterior fossa craniotomies might be necessary to
achieve
adequate exposure. Generally, the dura is a good barrier for tumor
spread
and it
must be removed only when necessary. If the dura is found infiltrated,
its
incision is undertaken and it is excised until free margins are reached.
If
the
facial nerve is invaded by the tumor, it should be included in the
specimen.
The
facial nerve may otherwise be re-routed to give access to the tumor. The
sigmoid
sinus and jugular bulb are preserved unless infiltrated. If there is any
area of
uncertain tumor clearance around the jugular bulb or the lower cranial
nerves it
is advisable to leave a vascular clip in situ for the site to be
identified
and
targeted during postoperative radiotherapy.

Fig 3: Subtotal Temporal Bone resection
Total temporal bone resection
This procedure is used in advanced T4 tumors. It may be performed with or without resection of the pinna. Bone is resected superiorly for 3 cm above the temporal line to expose the middle fossa dura and behind the sigmoid sinus by a similar amount to leave a residual margin of healthy bone. Medial dissection extends through the labyrinth and exposes the intrapetrous carotid artery. Inferiorly, the sigmoid sinus and jugular bulb are mobilized from surrounding bone. The sternocleidomastoid and digastric muscles are freed from the mastoid tip. At this stage in the procedure, the ascending ramus of the mandible is transected with a Gigli saw or a drill, and this and the head and coronoid process are dissected free and removed. A total parotidectomy is completed, and the specimen is removed en bloc. The residual tip of the petrous bone is then removed with a high-speed drill. Resection of the carotid artery can also be accomplished if the contralateral cerebral blood supply has been proven to be adequate by angiography and preoperative balloon occlusion.
Selection of surgical procedure.
The optimal surgical strategy for SCC of the temporal bone remains controversial. A review of the literature shows that many authors often adopt an individualistic approach to primary tumor and neck nodes management. Sleeve resection with retention of the pinna is still supported as a treatment of T1 tumors of the EAC . We do not perform SRs at our center where LTBR is the accepted mainstay for T1 and T2 tumors. Both LTBRs and subtemporal bone resections (STBRs) are accepted techniques in T3 tumors . In T4 tumors, although STBR is the surgery of choice, the application of total temporal bone resection (TTBR) in advanced T4 tumors is a matter of debate. The management of more advanced tumors (T4) is particularly challenging as a result of the complex anatomy and the proximity of intracranial structures. The palliative benefits of TTBR like decreased pain and improved hygiene in comparison with radiotherapy. Although recent studies reported an improvement in terms of surgical morbidity and survival in patients who have undergone TTBR, this procedure is still associated with significant postoperative deficits. STBR performed by a combination of en bloc and piecemeal resection techniques followed by postoperative radiotherapy could be a reasonable choice in patients with T4 tumors. Tumors of the EAC can involve the parotid gland either by direct extension or through nodal dissemination of the disease because one of the first echelon lymph nodes involved in SCC of the temporal bone is the intraparotid or periparotid node. Preformed pathways around the EAC like the cartilaginous fissures of Santorini, the petrosquamous suture line and the bony foramen of Huschke are suspected to facilitate easy spread of tumor anteriorly. This is the main reason why some surgeons prefer to associate superficial parotidectomy with LTBRs, specially if tumor is found to be eroding the anterior wall of the EAC. The incidence of parotid involvement in SCC of the EAC has been reported to be between 10 and 62% of patients . But there is no recorded benefit of performing a routine superficial parotidectomy in terms of better survival rates. It is well known that nodal dissemination in TBMs is uncommon and hence many authors avoid performing a routine superficial parotidectomy for the sake of nodal clearance . We do not include superficial parotidectomy as a routine procedure in all T2 cases and prefer to perform it in T2 tumors only if there is evidence of involvement of the anterior wall of the EAC. However, in T3 tumors, we routinely perform a superficial parotidectomy. In T4 tumors and when there is evidence of involvement of the parotid gland per se, we perform a total parotidectomy. Though lymph nodes involvement is relatively rare (10–36%) in TBMs, the presence of nodal metastasis, seen in advanced stages, has an adverse outcome on survival. Although some studies have reported a poor survival rate in the presence of nodal metastasis, others have not found this to be statistically significant. With this background it is difficult to justify a routine neck dissection in a N0 neck, specially in T1 and T2 tumors. A positive neck at presentation is a sign of aggressiveness of the primary tumor. Therefore, when there is clinical or radiological evidence of neck nodes involvement one must pursue an aggressive neck dissection. In N0 neck, we perform a frozen section of the level II lymph nodes and proceed with neck dissection only when this is positive for metastasis. In Nþ necks, we perform routine neck dissection to involve the parotid and the level II lymph nodes. Although radiotherapy has proven to be an effective adjuvant therapy, its role as a primary treatment modality for TBMs is not established. Postoperative radiotherapy is used to improve local and regional control of the disease. It is indicated in T3 and T4 tumors and also in T1 and T2 tumors where there is evidence of bone invasion, a positive margin, perineural invasion or nodal metastasis.
CONCLUSION
Despite the fact that significant attention has being focused on TBMs in the last few decades, there is still need to comprehensively study the entire gamut of histological varieties, refine the existing tumor staging system or develop novel ones and standardize treatment protocols. Radical surgery is the mainstay of treatment of TBMs in all tumor stages. Advances in neuroradiology, skull base surgical techniques and neuroanesthesia have made surgical resection of even advanced tumors in this area feasible with minimal morbidity. Adjuvant radiotherapy is indicated in T2, T3 and T4 tumors and is considered in T1 only in case of incomplete resection. `

Petrous Bone Cholesteatomas
Mar 11, 2022 (2 min read)An epidermoid cyst affecting the petrous portion of the temporal bone. It extends beyond the inner ear towards the center of the skull...
Petrous Bone Cholesteatomas
An epidermoid cyst affecting the petrous portion of the temporal bone. It extends beyond the inner ear towards the center of the skull base. Clinical presentation of Petrous Bone Cholesteatoma is often variable and misleading. It can present as a middle ear cholesteatoma, progressive or sudden facial nerve palsy, sensorineural hearing loss or a conductive hearing loss, secretary otitis media or intracranial complication. Pathophysiology This disease can be either acquired or congenital. Petrous Bone Cholesteatomas (PBC) are most of congenital. It is believed that the congenital cholesteatoma of petrous apex is caused by the resting squamous cell. The disease is described as congenital when it manifests itself without a past history of trauma or infection with intact and located behind the tympanic membrane. This disease is destructive in nature; most of PBC appear to be asymptomatic and appear to be innocuous keratin pearl. In severe conditions, they can enlarge and caused serious complications that include the ossicular destruction, base of skull complications and facial paralysis.
What are the symptoms?
The commonest presenting symptom is unilateral hearing loss, followed by facial paralysis/ weakness due to involvement of facial nerve. They can also present with vertigo, numbness of face, blurring of vision/ double vision. Rarely, intracranial complications causes seizures/ altered sensorium.
How is it diagnosed?
Otoscopic findings: · Primary acquired: central perforation with cholesteatoma debris or retraction pocket with squamous epithelium and debris
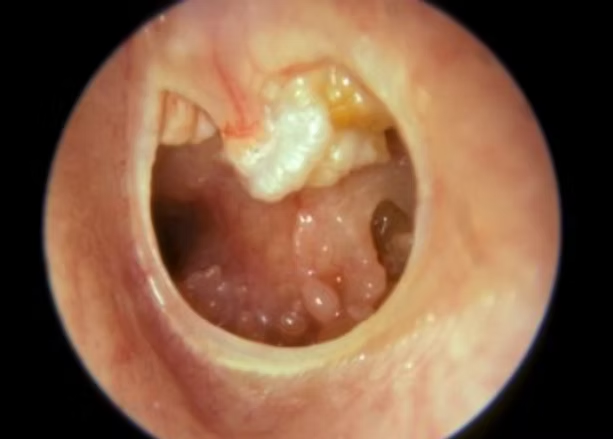
Fig 1(A): A case of chronic suppurative otitis media with central perforation and cholesteatoma seen in the middle ear cavity. (B): Attic retraction pocket with cholesteatoma.

Congenital and secondary acquired: white or pearly mass behind the intact tympanic membrane
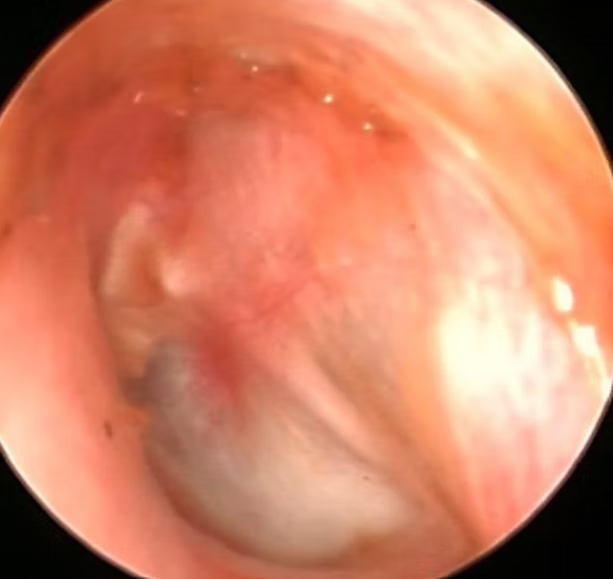
Fig 2: whitish mass behind the intact TM
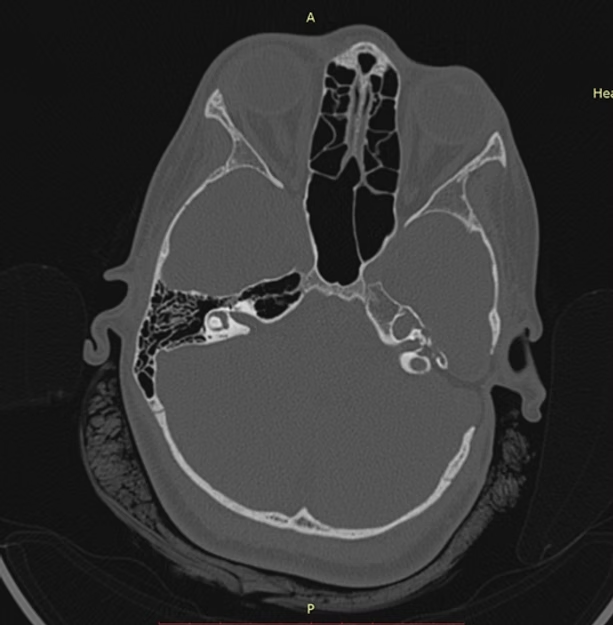
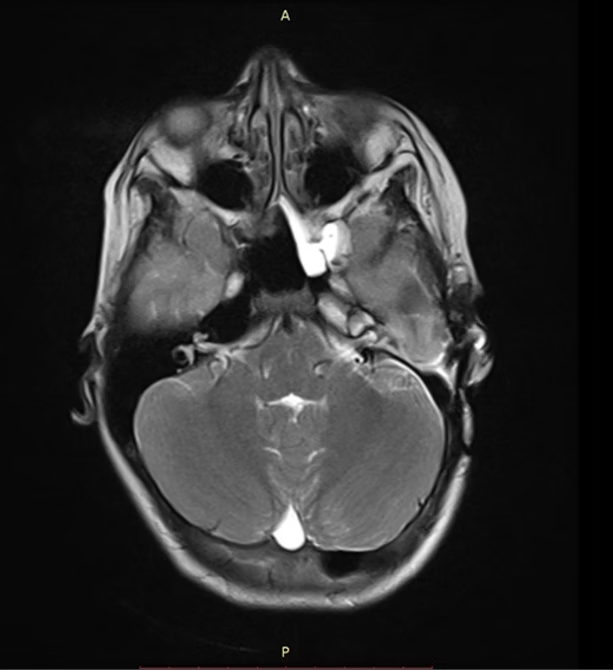
Fig 3: Diagnosis of Petrous bone cholesteatoma in CT and MRI scan.
Currently, the advances in radiology have greatly improved the detection and diagnosis of PBC. Computer tomography (CT) can clearly show the destruction of bone as well as the anatomic relationship between lesions and relevant surgical marks which will provide direct reference for making appropriate surgical plans. Magnetic resonance imaging (MRI) is more helpful to distinguish PBC from other soft tissue lesions, and the sequence of diffusion weighted imaging (DWI) is the best method to detect residual and recurrence of PBC.
Audiometry:
To assess the degree of hearing loss and to plan for hearing rehabilitation post-surgery.
Management of Petrous Bone Cholesteatomas
Surgery remains the cornerstone for treatment of PBC. The translabyrinthine-transcochlear approach, transotic approach, infralabyrinthine approach, and infracochlear approach represent the most direct surgical approaches. Translabyrinthine-transcochlear approaches are aggressive approaches that require sacrifice of any residual hearing with or without transposition of the facial nerve. The infralabyrinthine approach and infracochlear approach are hearing preservation surgeries. Post-surgery the cavity is closed permanently. Hence hearing rehabilitation can be done using various implantable such as bone anchoring hearing implants and cochlear implantation.
The middle cranial fossa approach, which may allow for hearing preservation, may be performed in patients with good hearing and normal facial nerve function, but requires a craniotomy, affords poor access, and may involve traction on important neurologic structures with potential for injury.
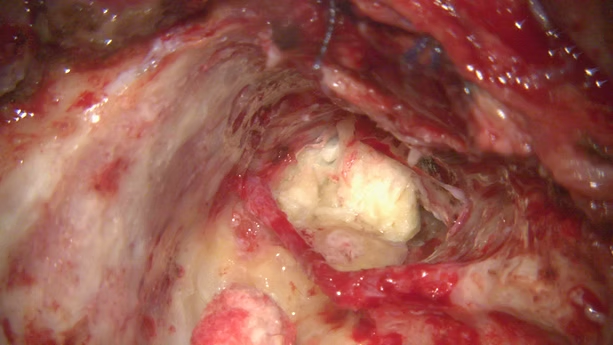
Fig4: Petrous bone Cholesteatoma seen intraoperatively
Fig5: Post-surgery OSIA for hearing rehabilitation

Recent therapeutic advances in Mucormycosis
Feb 9, 2022 (5 min read)Mucormycosis, a rare angioinvasive disease, has emerged as a serious opportunistic fungal infection. It spreads rapidly causing tissue...
Recent therapeutic advances in Mucormycosis
Mucormycosis, a rare angioinvasive disease, has emerged as a serious opportunistic fungal infection. It spreads rapidly causing tissue infarction and necrosis in post-COVID patients. Rhino-orbital-cerebral (ROC) presentation is the most common form.[1] Treatment includes extensive surgical debridement, high doses of systemic Amphotericin B (AmB), control of underlying disease and other supportive measures.
We emphasize that recent therapeutic advances have the potential to improve outcomes of Mucormycosis. Management strategies are based on recent preclinical and limited clinical data. Their validation requires definitive, prospective, and controlled clinical trials. Lipid formulations of amphotericin B (LFAB) have evolved as the primary therapy for Mucormycosis. Based on available data Posaconazole may be used as a salvage therapy but not recommended as primary therapy. Animal models and retrospective limited preclinical data have shown combinations of LFAB- echinocandin and azole-echinocandin therapy improve survival during Mucormycosis.[1] Immune stimulation due to enhanced exposure of β-glucan on the fungal surface and expressions of target enzyme for echinocandins by R. Oryzae, may be one of the mechanisms by which echinocandins improve outcomes. [2,3] However,combination LFAB-echinocandin therapy if considered for mucormycosis, echinocandins should be administered at US Food and Drug Administration-approved dosages. Combination of LFAB and the iron chelator also showed favourable results in animal models. However, the ongoing phase II, DEFEAT Mucor clinical trial failed to demonstrate a benefit of the combination regimen.[4] Ultimately, randomized, prospective phase III clinical trials will be required to determine whether combination therapeutic regimen is superior to monotherapy with an LFAB. Salvage therapy can also be considered with Granulocyte colony-stimulating factor-mobilized granulocyte transfusions which may provide additional support for persistently neutropenic patients Administration of granulocyte macrophage colony-stimulating factor or interferon-γ in nonneutropenic patients with refractory infection may further augment host response and antifungal effect. [1]
Radiological advances
Imaging modalities, such as CT and MRI with or without contrast, provide excellent structural resolution for visualizing advanced diseases, but generally are of limited value in detecting early disease. Therefore, for early diagnosis and treatment, metabolic and functional imaging techniques complement the role of anatomic imaging modalities for the optimal management of these patients. Limited studies have shown FDG, a nonspecific tracer has been found to accumulate at the sites of infection and can aid in detecting and localizing the exact sites of infection.[5]
The mechanism of increased FDG uptake in the inflammatory cells is due to upregulation of cellular glucose metabolism secondary to “respiratory burst”.[6] Study conducted by Hot et al, showed benefit of FDG PET in early diagnosis and staging of invasive fungal sinusitis. [7] One study conducted by Dumarey et al showed FDG-labelled autologous leukocytes more specific as neutrophils have more affinity to acute infections.[8] Another study proved FDG PET/CT is valuable in the early diagnosis and management of Mucormycosis with serial follow up (18)F-FDG PET/CT until complete patient recovery.[9] However more studies need to be done in proving the role of PET scan on early diagnosis and disease resolution post treatment.
Orbital Reconstruction
ROCM patients post orbital exenteration present with cosmetic deformity, persistent nasoorbital fistulas, which might need orbital reconstructions. A successful rehabilitation of anophthalmic socket needs a socket reconstruction with exenteration prosthetic fitting that mimics the contralateral globe. Various free flaps have been used to reconstruct the anophthalmic eye. Temporalis fascia is the most commonly used flap. Other flaps used are Radial artery free forearm flap, dorsalis pedis flap, gracilis free flap, latismus dorsi or rectus muscle free flap. However, flaps have limitations of its pedicle length, inability to provide bulk of tissue, donor site morbidity. Prosthetic rehabilitation with custom-made ocular prosthesis yields better and more satisfactory results both esthetically and psychologically as compared to a stock eye prosthesis. Advantages of custom-made ocular prosthesis is it retains shape of the socket, prevents accumulation of fluid in the cavity, maintains palpebral opening similar to natural eye.[5]
Transcutaneous retrobulbar injection of amphotericin B in rhino-orbital-cerebral mucormycosis
Several case reports have described transcutaneous retrobulbar injection of amphotericin B (TRAMB) and conservative orbital debridement with or without irrigation with amphotericin B as a viable option in halting orbital progression and providing as an opportunity to avoid orbital exenteration. Randomized clinical studies are required to prove the success rate of TRAMB, however it can be used as an adjunctive treatment with background of adequate sinus debridement.
Facio maxillary prosthesis
Rehabilitation of patients with faciomaxillary resection involving maxillae, hard and soft palate presents a significant challenge in restoring cosmetic deformity, speech, deglutition, mastication and respiration. Prosthodontic management using distraction osteogenesis, osseointegrated implants, bone grafts, and magnet attachments can be used to rovide support and stability of a large definitive obturator.[6]
Prosthodontic rehabilitation of patients with bimaxillary resection involving the maxilla, hard and soft palates, and paranasal sinuses presents a significant challenge in restoring speech, deglutition, mastication, and respiration. This clinical report describes the prosthodontic management of a young girl treated for leukemia who required a bilateral maxillectomy secondary to mucormycosis. Distraction osteogenesis, bone grafts, osseointegrated implants, and magnet attachments were used to provide retention, support, and stability of a large definitive obturator.
Hyperbaric oxygen therapy
Hyperbaric oxygen suppresses fungal growth in vitro and has theoretical value in treating mucormycosis because it reduces the tissue hypoxia and acidosis that accompany vascular invasion by the fungus.
References
1: Goldstein, Ellie JC, et al. "Recent advances in the management of
mucormycosis: from bench to bedside." Clinical Infectious Diseases 48.12
(2009): 1743-1751.
2: Ibrahim AS, Bowman JC, Avanessian V, et al. Caspofungin inhibits
Rhizopus
oryzae 1,3-β-D-glucan synthase, lowers burden in brain measured by
quantitative PCR, and improves survival at a low but not a high dose
during
murine disseminated zygomycosis, Antimicrob Agents Chemother, 2005, vol.
49
(pg. 721-7)
3. Lamaris GA, Lewis RE, Chamilos G, et al. Caspofungin-mediated
β-glucan
unmasking and enhancement of human polymorphonuclear neutrophil activity
against Aspergillus and non-Aspergillus hyphae, J Infect Dis, 2008, vol.
198
(pg. 186-92)
4. Spellberg, Brad, et al. "The Deferasirox–AmBisome Therapy for
Mucormycosis (DEFEAT Mucor) study: a randomized, double-blinded,
placebo-controlled trial." Journal of Antimicrobial Chemotherapy 67.3
(2012): 715-722.
5. Debnath N, Gupta R, Meenakshi A, Ramkumar K, John J. A Simplified
approach to fabricate a custome-made ocular prosthesis. Int J
Prosthodont
Restor Dent. 2013;3(1):25–29. doi: 10.5005/jpjournals-10019-1071 .
6. Oh, Won-suck, and Eleni Roumanas. "Dental implant–assisted prosthetic
rehabilitation of a patient with a bilateral maxillectomy defect
secondary
to mucormycosis." The Journal of prosthetic dentistry 96.2 (2006):
88-95.
7. Zhuang H, Alavi A. 18-Fluorodeoxyglucose positron emission
tomographic
imaging in the detection and monitoring of infection and inflammation.
Semin
Nucl Med 2002; 32:47–59
8.Bleeker-Rovers CP, Vos FJ, Corstens FH, Oyen WJ. Imaging of infectious
diseases using [18F] fluorodeoxyglucose PET. Q J Nucl Med Mol Imaging
2008;
52:17–29
9.Hot A, Maunoury C, Poiree S, et al. Diagnostic contribution of
positron
emission tomography with [18F]fluorodeoxyglucose for invasive fungal
infections. Clin Microbiol Infect 2011; 17:409–417 [Crossref] [Medline]
[Google Scholar]
10. Dumarey N, Egrise D, Blocklet D, et al. Imaging infection with
18F-FDG-labeled leukocyte PET/CT: initial experience in 21 patients. J
Nucl
Med 2006; 47:625–632
11. Liu, Yuejian, et al. "Utility of 18F-FDG PET/CT in diagnosis and
management of mucormycosis." Clinical nuclear medicine 38.9 (2013):
e370-1.

Current perspectives in the management of Tympanojugular paragangliomas
Sep 17, 2021 (9 min read)Tympanojugular paragangliomas, also commonly known as glomus tympanicum tumors, are benign slow growing tumours. Because of its indolent...
 Dr.
Sampath
Chandra Prasad Rao
Dr.
Sampath
Chandra Prasad Rao
Current perspectives in the management of Tympanojugular paragangliomas
Tympanojugular paragangliomas, also commonly known as glomus tympanicum tumors, are benign slow growing tumours. Because of its indolent nature, diagnosis is usually delayed until it reaches in significant size. They arise from rests of paragangliar tissue contained in the middle ear space. Although variations have been reported, these tumours most commonly arise from the dome of the jugular bulb: the tympanic branch of the gloss pharyngeal nerve (Jacobson’s nerve), the auricular branch of the vagus nerve (Arnold’s nerve) and a promontory mucosa. Paths of a spread of glomus jugulare tumours are predictable, following lines of a least resistance, including mastoid air cells tracts, vascular channels and jugular vein lumen, the Eustachian tube and neural foramina. The floor of the tympanic cavity is often destroyed by a superiorly spreading tumour, with a subsequent involvement of middle ear ossicles, destruction of the adjacent carotid crest and the jugular spine. Medial spread from the mesotympanum involves the cochlea; first, the tumour fills in intracochlear spaces, then causing osteonecrosis of the osseous labyrinth during a later stage.
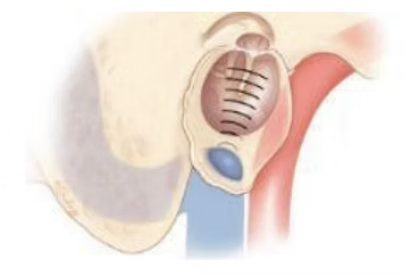
Clinical features
The most common presenting symptom for TJPs is hearing loss that is present in approximately 60-80% of cases with many subjects also complaining about their pulsatile tinnitus. The other symptoms include hoarseness, dizziness, or dysphagia. Facial paresis is occasionally present. They also typically present with otoscopic finding of a retro tympanic vascular mass. A history of palpitations and headaches is suggestive of catecholamine excess but is extremely rare for head and neck paragangliomas. Catecholamine excess is found in the familial etiology group due to the concomitant presence of a pheochromocytoma in the paraganglioma syndromes. Since catecholamine excess is rarely found in head and neck paraganglia, in case it is discovered, it should be assumed to be from a synchronous thoracic or abdominal lesion unless proved otherwise.
Fig 1: Retrotympanic reddish mass
Investigations
High-resolution computed tomography (HRCT) and T1-weighted (T1W), T2-weighted (T2W), and T1-weighted gadolinium-enhanced MRI sequences are necessary for identification of the suspected pathology as also the extent of the lesion. HRCT scan will show a moth-eaten appearance of the bone of the jugular fossa and erosion of the caroticojugular spine. Paraganglia give a low to intermediate signal on T1W and a high signal on T2W sequences. The salt and pepper appearance seen on T2W images is due to intratumoral vasculature appearing as flow voids. The scans give adequate information about the likely pathology and thus no biopsy is required for confirmation of the diagnosis. In the case of any suspicion, a 24-hour urine sample is analyzed for epinephrine, dopamine, and vanillymandelic acid. If elevated, 1,231-metaiodobenzylguanidine (mIBG) scintigraphy and MRI of the chest and abdomen may be used to localize the additional extracranial lesion.

Fig 3: HRCT scan showing moth-eaten appearance of the bone of the jugular fossa and erosion of the caroticojugular spine
Management
Based on the scans, the lesion is staged. Staging for glomus was popularized by Fisch and modified by Sanna. The current therapeutic approaches towards tympanojugular paragangliomas include surgical resection, irradiation, steretotactic radiosurgery with a Leksell gamma knife, or combinations of all modalities.
Surgery
The aim of the surgery is a radical removal of the tumour with a minimal morbidity. Various surgical approaches and treatment modalities has been discussed in the literature regarding large tumous. Radical treatment of these tumours is possible due to microsurgical and complex techniques, including preoperative embolization. Nowadays, the tumour control is achieved in 80–90% in most surgical series. To study the vascular supply of the glomus tumor, angiography is performed prior to superselective embolization of the feeder vessels. The embolization is preferred around 48 h prior to surgery, commonly using polyvinyl alcohol as the embolic material. Devascularization of the glomus tumor reduces its size, makes it firmer in consistency and decreases blood loss during surgery. Embolization of feeders from the external carotid artery and its ascending pharyngeal branch is routinely performed.
Conservative surgery
For small C1 tumors with minimal jugular bulb involvement, the hypotympanic approach has been described. A transjugular approach with preservation of the facial nerve may be used for C1 glomus jugulare tumors with a predominantly posterior or an intracranial extension. Preservation of the posterior canal wall in class C tumors invites the risk of leaving behind residual tumor. Hence, in class C tumors, the recommendation is to perform a "canal wall down mastoidectomy" with drilling of the tympanic bone, excision of the styloid process, and obtaining a good exposure and control of the internal jugular vein, ICA, and lower cranial nerves in the jugular foramen and neck. All these steps are incorporated in the infratemporal fossa A approach described by Fisch.
Surgical technique of the infratemporal fossa A approach for exposure of the jugular foramen
A C-shaped incision is taken from the top of helix in the midtemporal
area,
2.5-3 cm behind the postaural groove upto the mastoid tip, coursing two
finger-breadths below the angle of mandible up to the greater cornu of
the
hyoid bone. The skin and subcutaneous layers are elevated and reflected
anteriorly. The neck flap is elevated in the subplatysmal plane. A
rectangular-shaped musculoperiosteal layer is created just posterior to
the
external auditory canal, which helps in a two layered cul-de-sac closure
of
the external auditory meatus. The external auditory canal is transected
circumferentially at the level of the spine of Henle. The cartilage is
excised from this lateral half of the transected external auditory canal
till a circumferential skin cuff is created which is everted and sutured
as
the first layer of cul-de-sac closure of the external auditory canal.
The
rectangular musculoperiosteal layer created earlier is used as a second
layer closure. The skin of the medial half of the external auditory
canal
together with the malleus and the incus (after dislocation of the
incudostapedial joint) is excised. The greater auricular nerve that
courses
along the lateral surface of the upper part of the sternocleidomastoid
muscle is preserved or harvested. The anterior border of the
sternocleidomastoid muscle is separated from its covering cervical
fascia,
and its attachment to the mastoid is divided. This helps to retract the
sternocleidomastoid muscle and the temporalis muscles posteriorly. The
ICA,
external carotid artery, and the internal jugular vein are identified,
and
silicone tubing is loosely placed around them for later control if
needed.
The IX, X, XI, and XII cranial nerves are identified. The facial nerve
distal to the stylomastoid foramen is delineated including its distal
upper
and lower divisions in the parotid gland.
"canal wall down mastoidectomy" is performed. The mastoid tip is
excised.
The facial nerve is decompressed in 270 degrees circumference from the
geniculate ganglion till the stylomastoid foramen area. At the
stylomastoid
foramen area, the periosteum is left around the facial nerve. The facial
nerve is lifted off its bed and freed from the geniculate ganglion till
the
distal upper and lower divisions in the parotid . The facial nerve is
then
transposed anteriorly by lodging it in the parotid tissue and suturing
the
parotid tissue over the nerve so as to create a tunnel for the facial
nerve.
The tympanic bone is drilled off. The styloid process is exposed by
dissecting the muscles off it and excised. This exposes the underlying
ICA.
The bone over the sigmoid sinus and jugular bulb is taken off to expose
the
tumor over the jugular bulb. However, a small shelf of bone is kept
intact
over the upper end of the sigmoid sinus to facilitate its extraluminal
packing. The infracochlear cells are drilled off. The drilling can
extend
anteriorly to expose the infratubal vertical segment of the ICA when
desired
based on the tumor spread. In type C3 tumors, the infratemporal B
approach
is used. In this approach, the anterior canal wall is drilled off, and
the
temporomandibular joint is exposed. Drilling proceeds anteriorly from
the
base of zygoma. The middle meningeal artery and mandibular nerve are
transected. The horizontal petrous segment of the ICA is exposed along
with
the tumor over it. Drilling can proceed anteriorly until the foramen
lacerum
where the ICA is seen to course superiorly along its paraclival segment.
In
case the tumor has destroyed the cochlea and is infiltrating toward the
petrous apex, a transcochlear approach is performed. When the tumor
extends
into the sphenoid sinus or the nasopharynx, these need to be opened up.
This
forms the basis of infratemporal fossa C approach. In tumors extending
posteriorly from the jugular foramen, a far or extreme lateral approach
is
performed depending on the tumor spread.
Fig4: Infratemporal Fossa Type A approach: tumour clearance with IJV ligation and Sigmoid sinus extraluminal packing and anterior rerouting of facial nerve.
Management of the facial nerve
Management of the facial nerve that appears involved has been clearly defined by Fisch. In cases of grade I involvement (tumor is 1mm or more from the perineurium), the tumor bulk is gently dissected away from the facial nerve epineurium. In grade II involvement (epineurium invaded), the epineurium is resected leaving the perineurium intact. When the perineurium (grade III) or endoneurium (grade IV) is invaded, the nerve needs to be resected and an end-to-end anastomosis or an interposition nerve grafting is performed between the two cut ends of the facial nerve. Grafting is performed using greater auricular or sural nerve grafts. Rarely, in cases where grafting is not feasible due to extensive involvement of the proximal segment of the facial nerve, a second stage facial-hypoglossal anastomosis is an option.
Management of ICA
In cases with proven involvement of the wall of the ICA, a preoperative balloon occlusion test is mandatory. The surgical options include sacrifice of the involved ICA (with bypass, when the cerebral cross-circulation is inadequate) or its preoperative stenting. Preoperative stenting is nowadays preferred to the carotid artery sacrifice. Preoperative stenting of the ICA also facilitates removal of the tumor closer to its lumen without the risk of its rupture.
Management of the sigmoid sinus and the jugular bulb
A thin shelf of bone is left over the upper end of the sigmoid sinus. This is to facilitate its extraluminal compression to control venous bleeding during surgery. An extraluminal compression is the preferred option to control venous bleeding than intraluminal compression as the latter is associated with the possible risk of embolization as well as the risk of retrograde thrombosis of the transverse sinus and the vein of Labbe.
The internal jugular vein is ligated in the neck. It is dissected off the surrounding tissues and passed under the accessory nerve toward the jugular foramen. The lateral wall of the sigmoid sinus is excised along with the tumor and the internal jugular vein. At this point, brisk bleeding ensues from the openings of the inferior petrosal sinus located in the medial wall of the jugular bulb. These openings are meticulously packed with surgicel without any undue pressure since the lower cranial nerves traverse beneath the medial wall of the jugular bulb. Bleeding also arises from the posterior condylar vein, which is also controlled with surgicel. Complete ligation and excision of the sigmoid sinus is avoided as requires an additional dural patch to prevent a cerebrospinal fluid leak.
Intradural extensions
Small intradural extensions may be excised along with the involved dura in a single stage. However, in larger defects, it is not always possible to plug the defect securely. The opening of the fascial planes in the neck and extensive bony drilling further prevents a watertight dural closure. It is easier to seal dural defects in the second stage due to a smaller area of exposure and prior closure of communication with the fascial spaces in the neck.
Tumor involving the vertebral artery
Posteriorly based paragangliomas may involve the vertebral artery, lower clivus, and foramen magnum and require a far lateral or extreme lateral transcondylar approach for adequate exposure.
Role of Gamma Knife Radiosurgery
Stereotactic radiosurgery, by inducing vascular endothelial damage, may be particularly effective for vascular tumours, such as a glomus. Surgery is associated with higher morbidity rates but has the advantage of immediately and eliminating the tumor. The results of radiosurgery are promising; however, the incidence of long-term recurrence is at present not well defined. There is a consensus that surgery is the preferred option in young patients, in those with a catecholamine secreting paraganglioma, and in those with rapidly progressive neurological deficits. As the target volume of the tumor increases, there is a proportional increase in the radiation dose on the surrounding normal tissues. For this reason, stereotactic radiosurgery is not suited for large lesions. However, for residual or small recurrent lesions, stereotactic radiosurgery is very effective.
Conclusions
Management of glomus jugulare tumors requires a good knowledge of the
temporal bone and cervical as well as intracranial anatomy to evaluate
the
extent and progression of the tumor and the type of surgical approach
required.
Surgery is the preferred option in the young and in medically fit middle
aged to elderly patients in whom total extirpation of the tumor is the
aim.
Extensive infiltration of lower cranial nerves or the dominant vessels
is an
indication for a more conservative excision of the lesion. Improved
surgical
techniques have considerably decreased surgical morbidity. Speech and
swallowing therapy as well as extensive physiotherapy have helped to
overcome the morbidity arising due to lower cranial nerve paresis or
palsy.
In the elderly and medically unfit subjects with minimal symptoms, a
close
observation is an option. These subjects, if significantly symptomatic
or
showing disease progression, may be subjected to stereotactic
radiosurgery.
Stereotactic radiosurgery is the preferred option in residual or small
recurrent tumors.

Vascular loops at the Cerebellopontine angle: Is there a correlation with tinnitus?
Aug 20, 2021 (4min read)Tinnitus, sensorineural hearing loss (SNHL), and vertigo are common audio-vestibular symptoms and they are well-known classic triad in...
 Dr.
Sampath
Chandra Prasad Rao
Dr.
Sampath
Chandra Prasad Rao
Vascular loops at the Cerebellopontine angle: Is there a correlation with tinnitus?
Tinnitus, sensorineural hearing loss (SNHL), and vertigo are common audio-vestibular symptoms and they are well-known classic triad in inner ear disease involving the membranous labyrinth . Tinnitus it can be classified as pulsatile and non-pulsatile or objective and subjective. Pulsatile tinnitus is less common than non-pulsatile and can be due to vascular tumour such as glomus or vascular abnormality. AICA loops in the cerebellopontine cistern have been implied in causing auditory and vestibular symptoms, as well as hemifacial spasm, resulting from compression of the VII and VIII cranial nerves.
The vessels can be classified according to their anatomic location: type I: lying only in the CPA, but not entering the internal auditory canal (IAC) type II: entering, but not extending >50% of the length of the IAC type III: entering and extending >50% of the length of the IAC It is important to evaluate the presence of vascular contact and the angulation of eighth cranial nerve at the contact point, as specific signs of vascular compression. It has been proposed that compression of the vestibulocochlear nerve (8th cranial nerve) by a vascular loop of the anterior inferior cerebellar artery (AICA) could be the causative factor resulting in the otologic symptom. This pathology is described as vascular compression syndrome (VCS) which is caused by direct contact between a blood vessel and a cranial nerve. Initially, the hypothesis of VCS was suggested by McKenzie in 1936 and later, discussed by Jannetta in 1975, to refer to cranial nerve dysfunction.[1]
Vestibulocochlear compression syndrome
Microvascular compression of the vestibulocochlear nerve is known to cause disabling tinnitus and vertigo. Abnormal ABR, brief spells of vertigo, unilateral sensorineural hearing loss, abnormal vestibular findings, continuous tinnitus, hearing loss, abnormal electronystagmogram, and other findings have been reported to be diagnostic for neurovascular compression of the eighth cranial nerve.

Microscopic view of the longitudinal loop that the small branch from the AICA made around the facial nerve.
Pathophysiology
Various explanations were assumed to explain the impaired nerve’s function as an effect of vascular compression. Early in 1945, Sunderland et al. assumed that the proximity between the AICA and the nerves within the narrowed space of the IAC possibly produce nerve conduction disturbance due to the applied mechanical pressure via atheromatous, tortuous, or pulsating vessels [2]. The pulsatile vascular compression may result in nerve demyelination and/or fixation of the artery to the nerve by arachnoid adhesions [3]. It was also proposed that the arterial elongation and brain “sag” related to the aging process may result in cranial nerve cross-compression in the CPA [4]. Impaired blood flow through the vascular loop as a direct result of neurovascular compression was suggested to result in reduced vascular perfusion of the cochlea and vestibule leading to dysfunction [5].
An improvement in dysfunctional hyperactivity of the 8th cranial nerve was detected after microvascular decompression, which favored relation to the existence of a vascular loop .[5] Though the concept of vascular compression has been adopted for hemifacial spasm and trigeminal neuralgia, contradictory results have been reported about the relationship between VCS and neuro-otologic symptoms .[6]
Investigations
Highly sensitive MRI techniques have made it possible to investigate the relationship between intracranial vessels and nerves in a non-invasive manner. Volumetric sequences with strong T2 weighting (constructive interference in steady state imaging, fast imaging employing steady-state acquisition, and balanced fast-field echo imaging) present advantages over conventional angiographic examinations, given that, in addition to being non-invasive.
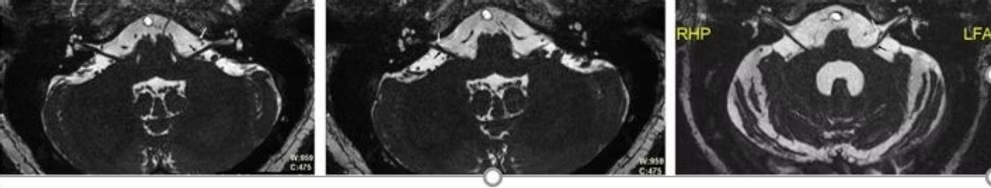
Different types of vascular contact in CISS images of the CPA and IAC with white arrows pointing to the vascular loop and the black arrows pointing to the corresponding 8th cranial nerve. (a) no contact, (b) direct contact, and (c) direct contact with mild angulation
Treatment
Microvascular decompression (MVD) of the vestibulocochlear nerve has been reported to be an efficient treatment option with high success rates of up to 80% for several vestibulocochlear compression syndromes. MVD for the vestibulocochlear nerve has successfully been performed for vessels compressing the nerve over its entire segment in the cerebellopontine angle. Several studies reported a complete recovery or marked improvement of subjective symptoms in 100% of the patients with vertigo and 65.5% of the patients with tinnitus. However, it is still difficult to consider neurovascular compression of the eighth cranial nerve as a major cause of disabling vertigo and tinnitus.
References
1.Jannetta PJ (1975) Neurovascular cross-compression in patients with
hyperactive dysfunction symptoms of the eighth cranial nerve. Surg Forum
26:467–469
2.Applebaum EL, Valvassori GE (1984) Auditory and vestibular system
findings
in patients with vascular loops in the internal auditory canal. The
Annals
of otology, rhinology & laryngology Supplement 112:63–70
3.Jannetta PJ (1980) Neurovascular compression in cranial nerve and
systemic
disease. Ann Surg 192:518–525
4.Brunsteins DB, Ferreri AJ (1990) Microsurgical anatomy of VII and VIII
cranial nerves and related arteries in the cerebellopontine angle.
Surgical
and radiologic anatomy : SRA 12:259–265
5.Jannetta PJ (1997) Outcome after microvascular decompression for
typical
trigeminal neuralgia, hemifacial spasm, tinnitus, disabling positional
vertigo, and glossopharyngeal neuralgia (honored guest lecture). Clin
Neurosurg 44:331–383
6.Nowe V, De Ridder D, Van de Heyning PH, Wang XL, Gielen J, Van Goethem
J
et al (2004) Does the location of a vascular loop in the
cerebellopontine
angle explain pulsatile and non-pulsatile tinnitus? Eur Radiol
14:2282–2289

Skull Base Osteomyelitis: An emerging clinical entity.
Apr 23, 2021 (3min read)What is Skull base osteomyelitis (SBO)? Skull base osteomyelitis (SBO) is a complex and fatal clinical entity that is often misdiagnosed...
 Dr.
Sampath
Chandra Prasad Rao
Dr.
Sampath
Chandra Prasad Rao
Skull Base Osteomyelitis: An emerging clinical entity.


What is Skull base osteomyelitis (SBO)?
Skull base osteomyelitis (SBO) is a complex and fatal clinical entity that is often misdiagnosed for malignancy. SBO is commonly a direct complication of otogenic, sinogenic, odontogenic, and rhinogenic infections. It is an infection of the temporal, sphenoid, or occipital bone that can be a challenge to diagnose because of its nonspecific symptoms, long clinical course, and radiologic findings that mimic those of other entities.
Clinical profile
Patients with SBO frequently have nonspecific symptoms such as headache and facial pain, and therefore they exhibit a variety of clinical presentations. Most patients start with otitis externa and present with a headache, severe otalgia, facial pain, purulent otorrhea, and, in advanced cases, conductive hearing loss. In other cases, the origin might be a sinus disease, resulting in nasal congestion, rhinorrhoea, headache, and fever. Various cranial nerve palsy is also common.In general, patients usually experience lower cranial nerve palsy, because the inflammatory process is near the neural foramina. Because of its proximity to the external auditory canal at the exit of the skull base, one of the nerves that is most frequently affected is cranial nerve VII when the stylomastoid foramen or the mastoid process are involved, and this can cause facial pain and peripheral facial paralysis. This condition can be accompanied by sensorineural hearing loss when cranial nerve VIII is involved. Abducens nerve (VI) palsy can occur if the petrous apex is involved, manifesting as binocular diplopia. If the jugular foramen is affected, palsy can be present in cranial nerves IX, X, and XI and can cause a soft palate mobility disorder, unilateral vocal cord paralysis, and an inability to raise the arm above the horizontal plane, respectively. Similarly, hypoglossal canal involvement causes cranial nerve XII neuropathy, with tongue movement disorders.
Who are at risk?
Patients with SBO typically are elderly and have diabetes or are immunosuppressed. Other diseases that alter the vascularization and oxygenation of bone predispose patients to SBO, such as small vessel disease, radiation exposure, malignancy, osteoporosis, osteopetrosis, anaemia, malnutrition, and Paget disease of the bone. Other risk factors include cardiovascular disease, renal failure, hepatic failure, obesity, smoking, extended hospital stays, and chronic pulmonary disease. Causative pathogens Pseudomonas aeruginosa is the most prevalent bacteria, followed by Staphylococcus aureus. Other bacteria that are seen less frequently include Staphylococcus epidermidis, Salmonella species, Proteus mirabilis, nontuberculous Mycobacterium species, Streptococcus pneumoniae, Treponema pallidum, and Klebsiella species. There is general agreement that the most frequently seen fungus is Aspergillus species, although it depends on the study.
Investigational approach
The imaging techniques used to diagnose SBO are nonenhanced and contrast-enhanced CT, enhanced MRI, and nuclear imaging. CT is the best option for evaluating bone erosion and demineralization, MRI can help delineate the anatomic location and extent of disease, and nuclear imaging is useful for confirming bone infection with high sensitivity.
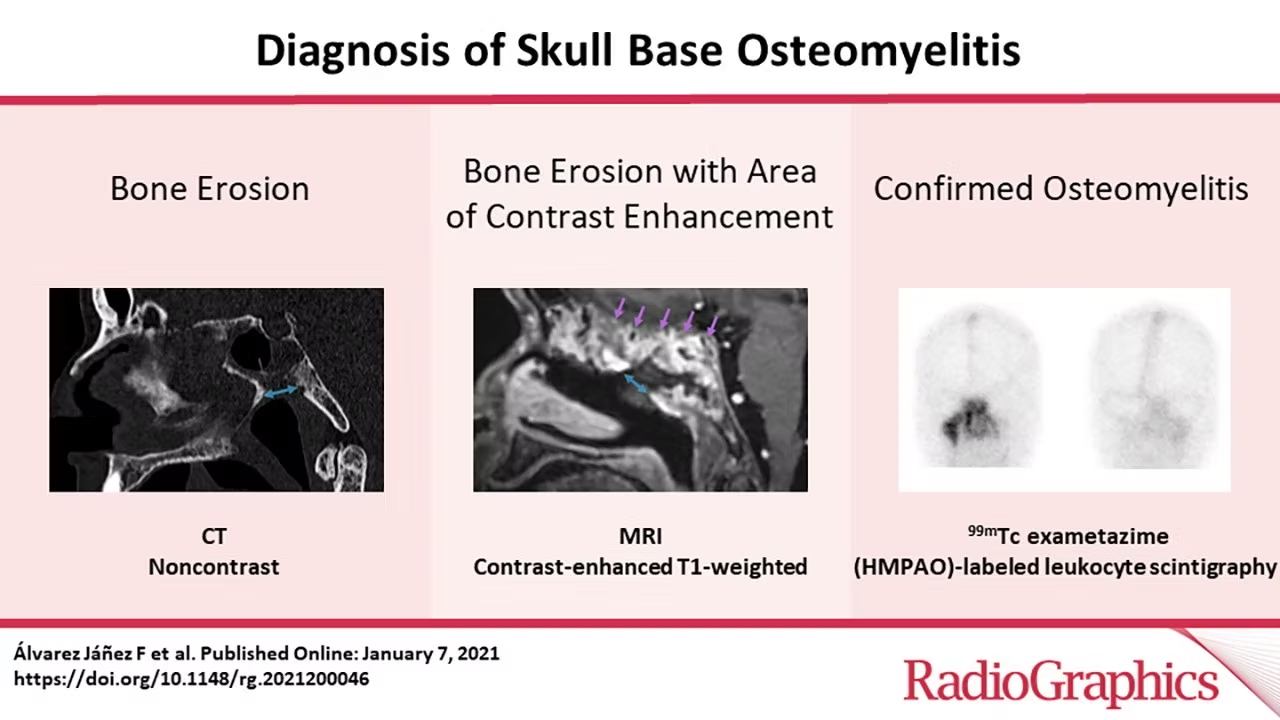
3:Coronal CT image shows the spread of infection (green arrows) in a patient with otogenic SBO of the temporal bone. SBO is usually secondary to otitis externa. From the external auditory canal, the infection reaches the temporal bone through the fissures of Santorini. The infection can spread anteroinferiorly to the parotid, masticator, or parapharyngeal spaces, medially to the carotid or perivertebral spaces, and even into the spinal canal. Intracranial spread can also occur by extension through the petroclival synchondrosis.
Management techniques
Complete resolution of the SBO cases may take several months. Since early
treatment can improve mortality rates, it is paramount that the
reporting
radiologists and treating clinicians are aware of the cardinal
diagnostic
signs to improve clinical outcomes of the disease.
Given the clinical suspicion for SBO and compatible radiologic imaging,
early empirical therapy with broad-spectrum intravenous antibiotics is
essential. Adequate control of risk factors (such as diabetes) is also
important. Intravenous antibiotics should be administered for at least 6
weeks, with subsequent oral antibacterial or antifungal therapy for
12–48
weeks.
Surgical debridement might be necessary in specific cases, including
those
with extensive soft-tissue involvement, severe pain despite antibiotic
treatment, complications (abscess formation), or bone sequestrum, as
well as
refractory cases. Other management approaches for complications, such as
endovascular therapy for pseudoaneurysms and anticoagulant therapy for
venous sinus thrombosis, should be individualized.
Combining hyperbaric oxygen therapy can improve the treatment, given
that it
increases the partial pressure of oxygen, reverses tissue hypoxia,
improves
phagocytosis, and promotes angiogenesis and osteoneogenesis. Treatment
consists of 100% oxygen administered for 90 minutes at 2.5 atmospheres
of
pressure 5 days a week for 1 month.
The global survival rates for SBO are approximately 90% at 18 months and
57%
at 3 years. In the diabetic population, the survival rates decrease by
21%–70%. Depending on the study, the mortality rate ranges from 9.5% to
46%.
The rate of neurologic sequelae is 31%–48%.
Reference
Álvarez Jáñez F, Barriga LQ, Iñigo TR, Roldán Lora F. Diagnosis of Skull
Base Osteomyelitis. RadioGraphics. 2021 Jan;41(1):156-74.
Prasad SC, Prasad KC, Kumar A, Thada ND, Rao P, Chalasani S.
Osteomyelitis
of the temporal bone: terminology, diagnosis, and management. J Neurol
Surg
B Skull Base 2014;75(5):324–331. Crossref, Medline, Google Scholar

State-of-the-art Audiology, Speech & Language Pathology Center at BSBI-BEIRC
Jun 6, 2020 (6 min read)In a broad sense, Audiology and Speech Language Pathology is the science and study of communication, hearing and balance. Audiology...
 Shamili Asokan
Shamili Asokan
State-of-the-art Audiology, Speech & Language Pathology Center at BSBI-BEIRC
State-of-the-art Audiology, Speech & Language Pathology Center at BSBI-BEIRC
In a broad sense, Audiology and Speech Language Pathology is the science and study of communication, hearing and balance. Audiology involves the study, assessment, and management of hearing, hearing loss, and balance related disorders while speech language pathology involves diagnosis and treatment of various communication and swallowing disorders. Although a loosely based understanding of hearing and speech problems was already existent, audiology and speech pathology as a requirement surged after World War II, when noise induced hearing loss and brain injury became a common issue among servicemen.
Who are Audiologists and Speech Language Pathologists and what are their roles as health care professionals?
An audiologist is a licensed professional who specializes in the diagnosis and management of hearing and balance disorders in all age groups, while Speech Language Pathologists (SLPs) specialize in the assessment and management of speech, communication and swallowing disorders, They are usually part of a larger multidisciplinary team that includes Otolaryngologists, Neurologists/Neurosurgeons, Pediatricians, Physiotherapists, Occupational Therapists, Teachers, Psychologists, and other healthcare professionals.
Hearing & Balance:
As a trained professional, audiologists have a comprehensive understanding of the auditory-vestibular systems, and its relevant anatomy and physiology. They possess extensive knowledge about the way sound is produced, transmitted, and perceived, which is crucial for aural habilitation and rehabilitation. For the assessment of hearing and balance, an audiologist employs various subjective and objective diagnostic tests in the form of a “test battery”. All individuals presenting with hearing and balance symptoms do not have to undergo every single test. The audiologist is required to select an appropriate test battery based on each patient’s individual complaints and health history. Once a conclusive diagnosis is made, the audiologist can provide management options, rehabilitation services, or make referrals to other appropriate professionals.
Types of Hearing Loss:
Hearing loss is usually classified as 3 types: Conductive hearing loss (hearing loss due to issues in the outer and middle ear such as outer ear malformations, ear infections, issues related to the middle ear ossicles, etc.). Sensori-neural hearing loss (hearing loss caused due to problems in the inner ear or the auditory nerve). Mixed hearing loss (a combination of conductive and sensori-neural hearing loss).
Diagnostic Tests:
The most basic and most commonly used audiometric procedure is the Pure Tone Audiometry (PTA). The test makes use of single frequency tones or “pure tones” given via air and bone conduction pathways to determine the type and severity of hearing loss done across a range of frequencies. Tympanometry or Impedance Audiometry (IA) assesses the middle ear functioning and the mobility of the tympanic membrane in by varying the air pressure in the external auditory canal. Speech Audiometry checks the patient’s ability to identify and repeat sound stimuli at different sound intensities. The results of these tests in conjunction can identify majority of the problems related to the cause of hearing loss. Otoacoustic Emissions (OAE) and Brainstem Evoked Response Audiometry (BERA) are two objective tests that can be used for screening as well as diagnostic purposes across all age ranges, especially pediatric population. OAE tests the functioning of the cochlear hair cells, specifically the outer hair cells, in response to acoustic stimuli. BERA is a non-invasive procedure that uses electrodes placed on the mastoid and forehead areas to detect electrical activity from the auditory nerve to the inferior colliculus. Vestibular Evoked Myogenic Potential (VEMP) is a test for vestibular function done by stimulating the ear with high intensity acoustic stimuli and recording the muscle activity via electrodes. There are two types of VEMP – cervical VEMP (cVEMP) tests the saccule and inferior vestibular nerve, while the ocular VEMP (oVEMP) tests the utricle and superior vestibular nerve. Videonystagmography (VNG) measures involuntary eye movements known as nystagmus. VNG uses infrared goggles to track the eye movements during positional changes and a variety of visual stimuli. These tests are valuable tools that assist in the diagnosis of vestibular and balance related disorders such as Meniere’s Disease, Superior Semicircular Canal Dehiscence (SSCD), etc.

Aural Habilitation & Rehabilitation:
After an accurate diagnosis is made, the audiologist will counsel a patient regarding treatment options and make additional referrals to other required professionals. Due to advances in technology, there are a multitude of devices that can prescribed based on patient needs and presenting medical condition. One of the most conventional devices are the hearing aids that can benefit almost all types of hearing loss of varying degrees. There are a vast range of brands and models of hearing aids available today that can be programmed according to patient needs, lifestyle, etc., There are also surgical treatment options available for people who receive little to no benefit from amplification devices or are unable to wear them. Middle Ear Implants and Bone Anchored Hearing Implants are surgically implanted hearing devices that convert sound into vibrations to stimulate the middle ear ossicles and the functioning cochlea via bone conduction respectively. Cochlear Implants (CI) and Auditory Brainstem Implants (ABI) are more complex surgical implants. A cochlear implant uses electrodes inserted into the cochlea to directly stimulate the auditory nerve in cases where the inner ear is damaged. In cases where there are malformations or damages to the auditory nerve with or without inner ear problems, an ABI bypasses the cochlea and auditory nerve to directly stimulate the cochlear nucleus in the brainstem. Research has shown that therapeutic assistance in addition to early intervention is of crucial importance to develop speech, language, and communication skills in pediatric patients. Therapy involves improving auditory and listening skills of the child, as well as training family members to work with the child in different environments and to make maximum use of the hearing device. Adults mostly counselling regarding care and management of their own devices rather than extensive therapy in the absence of other speech or cognitive impairments.

Speech, Language, and Communication:
SLPs usually specialize in the aspect of speech production rather than perception, focusing on a range of communication and swallowing disorders. The problems that fall under the umbrella of speech language pathology include (i) speech disorders (which is a difficulty with the production of speech sounds) like stuttering, articulation difficulties, voice disorders, and motor speech disorders of neurological origin; (ii) language and communication disorders that manifest as a difficulty in understanding and using language in appropriate or socially adequate ways; (iii) cognitive disorders that are usually secondary to traumatic brain injuries, cerebrovascular strokes, and other degenerative disorders; (iv) swallowing disorders
Assessment and Management:
A complete assessment of speech and language disorders includes comprehensive documentation of patient history along with a wide range of diagnostic tools for an accurate diagnosis across all age ranges. SLPs are required to plan and develop extensive treatment plans based on the assessment as well as monitoring patient progress and adjusting plans accordingly.
Voice Therapy:
SLPs use multiple therapeutic approaches in order to develop an effective treatment plan that need to be tailored for individual needs. Voice therapy uses direct and indirect approaches based on the long-term and short-term goals. Direct approaches involve manipulation of vocal mechanisms for healthy voice production, whereas indirect approaches focus on modifying the environmental aspects such as couns elling and implementing healthy vocal hygiene.

Adult neuro-rehabilitation and Swallowing Therapy:
Adult speech and language rehabilitation depends on the type of acquired disorder. Cognitive-communication and speech disorders result in memory issues, slurred speech, weakness of facial and oral muscles, communication and listening difficulties secondary to higher neurological issues. In such cases, the goals may include strategies to prevent further degeneration of memory, tactics to improve coherent speech production and social communication, exercises to strengthen oral motor, and facial muscles. In severe conditions, SLPs may introduce aided communication known as Augmentative and Alternative Communication (AAC). Difficulty in swallowing is termed as dysphagia. Swallowing requires coordination of the mouth, tongue, palate and esophagus. If even one of these aspects do not function adequately, it causes a difficulty in swallowing. Before management begins, SLPs along with otolaryngologists conduct a comprehensive swallowing assessment including a basic swallow test and Fiberoptic Endoscopic Evaluation of Swallowing (FEES). SLPs will use a combination of compensatory strategies along with direct treatment to improve the swallow function. Compensatory strategies involve improving control over swallowing in order to prevent aspiration while direct treatments use exercises to improve overall range of motion, and strengthening of muscles.

Our Bangalore Skull Based Institute (BSBI) and Bangalore ENT Institute and Research Center (BEIRC) group has a full-fledged audiology and speech department, providing a comprehensive assessment and management of speech, hearing and balance disorders, including Pure Tone Audiometry, Tympanometry, OAE, BERA, VEMP, VNG, Voice and Swallowing Assessment and Management. BSBI-BEIRC-BCIC is a one of a kind, one stop solution for your speech and hearing requirements. To know more about our services visit https://www.beirc.net/ & at https://www.bsbi.info/

COVID 19 is here to stay. What is the future of ENT surgical practice?
Jun 5, 2020 (1 min read)Watch the recorded Webinar on “COVID 19 is here to stay. What is the future of ENT surgical practice?” with 6 following panelists: · Dr....
 Shamili Asokan
Shamili Asokan
COVID 19 is here to stay. What is the future of ENT surgical practice?
State-of-the-art Audiology, Speech & Language Pathology Center at BSBI-BEIRC
Watch the recorded Webinar on “COVID 19 is here to stay. What is the future of ENT surgical practice?” with 6 following panelists: · Dr. Subhaschandra Shetty, MS FRACS, New Zealand · Dr. Arunachalam Iyer, MS, UK · Dr. Chang Wai Tsz, MD, Hong Kong · Dr. Puya Dehgani-Mobaraki, MD, Italy · Dr. Chethan M K, MD, (Anaesthesiology), Abu Dhabi · Dr. Sampath Chandra Prasad Rao, MS, DNB, FACS, FEBORLHNS, FEAONO, India (as Moderator) See the programme live on Facebook at https://www.facebook.com/entuned.zydus.3


Post lockdown mantra: learn to live with the Coronavirus
Jun 5, 2020 (9 min read)Question on everyone’s mind right now: What will life be like when India and the rest of the world lift the lockdown completely? Will...
 Shamili Asokan
Shamili Asokan
Post lockdown mantra: learn to live with the Coronavirus
Post lockdown mantra: learn to live with the Coronavirus
Question on everyone’s mind right now: What will life be like when India and the rest of the world lift the lockdown completely? Will life as we knew it before Covid-19 return? Well answer is Time will heal and time will answer!!! Coming to history itself, the world has survived all the pandemics of the past. We are alive and more populous than ever is proof to that. Humans have vanquished past pandemics with much lower degree of medical advancements. We have advanced medical facilities now. Optimists hope the disease will peak and fall after June. If it continues as it is, most likely the best policy will be to ease curbs even in red and orange zones, along with precautions and behavioural changes. Once the storm is over, you will not remember how you made it through, how you managed to survive. You will not even be sure whether the storm is over. But one thing is certain. When you come out of the storm, you won’t be the same person who walked in. “Virtual partying” is a term that has gained currency over the past two months as a world in quarantine looked around for new ways to socialise, along with other words or phrases that have become commonplace, like “social distancing” and “Zoom bombing.” And, as we take uncertain steps into a not so brave new world after lockdown, another term will perhaps get even more commonplace. The “new normal,” the lockdown may have made us retreat into a bubble, but it is a digitised bubble, from virtual parties to conference calls, webinars to online learning courses.

TOUCH ME NOT
In the post-Covid-19 world, “no contact” might become the standard. The new normal of social distancing, masks, gloves and washing of our hands is here to stay. Even if all restrictions are lifted, until a vaccine is found, we must remember that the virus is still among us. So, unless we continue to follow social distancing norms, we are going to see a spurt in cases in India. Do expect identification of hotspots to continue and cluster shutdowns. There will probably also be temperature sensors at public places and quarantine measures will be put in place if an infection is detected in a workplace. Greetings of the people by shaking hands, consoling the patients by patting the back with warm expressions, all will stop. Examination and diagnosis by tactile sensation will be transformed to the further investigations for the diagnosis. Waiting periods for doctor will increase. Each patient consultation time in between will increase. Opd consultation fees will be hiked, procedure cost will raise. Building relationship between people will take a different mode. Middle seats won’t be sold in flights, every other seat will be empty. Bus or train where new a platform to observe different culture, different people, making friendship will go on silent mode.
HEAL THE WORLD:
The lockdown may have made us insular, but it also brought the world closer. We adapted to our forced new life in lockdown quickly. We went digital and took to technology, reaching out to the community outside our insular lives. And while we stayed locked inside our homes, the birds came back to our gardens, the air became cleaner and the sky clearer. The smog in most of our cities lifted, with no vehicles to spew toxic gases. Young working couples adapted to ‘Work from Home’ (WFH), home-schooled their children, and used video conferencing for work calls and family socials. Covid-19 is reminding us of a simple but vital truth: “we are one species, sharing one planet.”
POST CORONAVIRUS CIVILISATION:
Basic hygiene rules learnt during the times of coronavirus outbreak and lockdown must not be forgotten and thrown out of practice. It would be wise for everybody to carry something like a first-aid box all the time that contains a soap bar, a pair of face masks, a pair of hand gloves, a vile of sanitiser, and a note to remind about social distancing. It will be curious to see whether the “new normal” is just a bump along the way or a path-changing reality. William Shakespeare’s paraphrased poem best captures the nation’s dilemma. Unlike Prince Hamlet, this is not a question of one’s own life but a billion other lives. It is critical to make decision to live with corona on sound facts and evidence, not fear or hope. New evidence shows that such fear and panic are unwarranted and a gradual return to a new and safe normal is the best option. It takes tall political leadership to calm nations and lead people out of their deep fears. Just as President Franklin Roosevelt did in 1933 when he exhorted Americans to shed their fear of the great depression, with the words “There is nothing to fear but fear itself.” So, let us learn to live with corona.
LIVE AND FIGHT TOGETHER– BUT REMEMBER DIVIDED WE ARE SAFE AND CROWDED WE ARE DOOMED
It is clear the current strategy of shutting down large parts of society is not sustainable in the long-term. The social and economic damage would be catastrophic. Corona is not going to disappear. Keep washing hands, using tissues while sneezing, avoid touching the face, masks, gloves the habit should continue. People should get exposed to the virus to develop the herd immunity. Cities will lose part of their variety and public social life. There will be less eating out, more home delivery, and lower consumption of luxuries. Public cinemas will turn into home cinemas. Gyms and hair salons will not be in demand for quite some time, unless good practices of social distancing and hygiene are maintained
LET US FOOL OURSELVES NO LONGER:
What his exit strategy was "Long term, clearly a vaccine is one way out of this, and we all hope that will happen as quickly as possible." Vaccine research is taking place at unprecedented speed, but there is no guarantee it will be successful and will require immunisation on a global scale. The best guess is a vaccine could still be 12 to 18-months away if everything goes smoothly. That is a long time to wait when facing unprecedented social restrictions during peacetime. Best vaccine available right now is distancing yourself from virus.What his exit strategy was "Long term, clearly a vaccine is one way out of this, and we all hope that will happen as quickly as possible." Vaccine research is taking place at unprecedented speed, but there is no guarantee it will be successful and will require immunisation on a global scale. The best guess is a vaccine could still be 12 to 18-months away if everything goes smoothly. That is a long time to wait when facing unprecedented social restrictions during peacetime. Best vaccine available right now is distancing yourself from virus.
TELEMEDICINE HERE TO STAY IN A POST-COVID-19 WORLD???:
Telemedicine is being used by doctors to connect with patients, and by mid-level provider/health workers to connect patients with doctors without patients having to physically visit a hospital or clinic. Those fancy Hollywood movies which portrayed the experience of tele-consultation, 3D image transfer will be a virtual reality in post covid world. I won’t call it as teleconsultation we can say it’s a virtual patient doctor interaction platform. Even post lockdown, it will help reduce the burden on the secondary hospitals and improve documentation, data-collection, diagnosis and care without risking the safety of the patients or the health workers. For a population of 1.36 billion, the doctor-population ratio 1:1,457, which is lower than the WHO recommended norm of 1:1,000. In addition to doctors, India has a little more than two million registered nurses and midwives, many of whom need infection control training to care for patients with communicable diseases, such as Covid-19 and tuberculosis. “Mobile apps, telemedicine, digital health are all great and welcome, but cannot help any country leapfrog fragile and under-resourced health systems, Telemedicine can increase reach, but it cannot replace a strong primary health system, which is dependent on competent health systems and providers. “I hope the biggest issue with public health delivery post-Covid is the recognition that universal health coverage is critical for any country to face a pandemic. Many countries are learning that you cannot build healthcare capacity during a crisis. It takes time and investment to build a good public health system. The COVID-19 pandemic brought telemedicine into a new light. As medical professionals need to stay healthy and disease-free, the need for remote technologies skyrocketed. Both the CDC and WHO are advocating for telemedicine to monitor patients and reduce risks of them spreading the virus by traveling to hospitals. A recent paper from The American Journal of Managed Care discussed the need for and challenges to incorporating telemedicine amidst the COVID-19 pandemic. It concluded that “although certain legal, regulatory, and reimbursement challenges remain, the COVID-19 outbreak may be the right impetus for lawmakers and regulatory agencies to promulgate further measures that facilitate more widespread adoption of telemedicine.”
ROBOTS TO THE RESCUE:
As it has been determined that the COVID-19 can spread from human to human, medical staff are at high risk of being infected. However, impervious to cross-infections are medical robots. These can be real game changers in cases of viral outbreaks. We have seen one such case already. where a man, diagnosed with the novel coronavirus, is being treated by a robot. The latter allows physicians to communicate with the patient via a screen and it is also equipped with a stethoscope, helping doctors take the man’s vitals while minimizing exposure to the staff. True, it won’t be possible in a jam-packed hospital in India with hundreds of such patients, but with time, quarantined patients could be better monitored with the help of robots. we believe that in the future, artificial intelligence could speed up drug creation and the clinical testing procedure through redesigning existing medicines or pairing unique ingredients virtually that were never tested before. For example, many pharmaceutical companies use supercomputers that root out therapies from a database of molecular structures. In 2015, biotechnology companies launched a virtual search for safe, existing medicines that could be redesigned to treat the virus. They found drugs predicted by the company’s artificial intelligence technology which may significantly reduce infectivity. This analysis, which typically would have taken months or years, was completed in less than one day. The five things we learnt from being isolated over the last two months: 1.Health: The idea that unequivocally matters the most is health. The impairment of health is an equal if not a bigger nightmare emotionally, logistically, and financially for the people around me. 2.Family: The first concentric circle of support at the end of it all is one’s family and young one needs older one’s shoulder and older ones need younger one hands. ‘Cats in the cradle’ typified this. When the child grows up, and the situation changes from a busy father neglecting his son to a busy son neglecting his father and both neglecting health for wealth. 3. Unfettered Social Media and its Evils: It is the reengineering by tech companies of the connected populace through drip dopamine hits with a significant tilt towards a profit motive of the tech companies rather than a social largesse of the populace. Social media thrives on the underlying human addiction- desire to get noticed. 4. Unsung heroes: Who would have imagined that doctors and health care workers will be in a battlefield with invisible enemy, but it’s a reality check to every nation about health infrastructure far more important than you border military service. The trench dwellers are usually the unsung heroes in any battle, be it the infantry in war or healthcare workers in a pandemic outbreak. Mankind has always had a steady state line of hands battling larger problems on the fringes of future-doctors, nurses, policemen, food delivery guys, Scientists, innovators, and researchers. These people have constantly worked towards pushing the boundaries of human capabilities significantly higher than the collective sum of our thinking would otherwise have 5. The world will not end, but the species will: The planet as we know it has been around for 4.5 billion years. We, for all our glorious achievements, have been around for only 300,000 years. The average life expectancy of the mammalian species is a million years. Abound 99 per cent of the species have gone extinct. We certainly have the capability and the mad dash to be a part of that statistic. Hope we wake up now and save ourselves going extinct.
FINAL CONCLUSION: HOPE HUMANS LEARN THEIR HUMANITY AND VASUDHAIVA KUTUMBAKAM (THE WORLD IS ONE FAMILY):
After the pandemic, hopefully, a new norm emerges— one where we share information across countries, collectively coordinate resources and work jointly for the survival of human race. And more importantly, live on a planet where we do not mess with nature. We are no longer a species that hunts to survive. We are one that evolved into one that breeds in captivity to kill and kills in masses to relish life. Hopefully, we find the balance and make sure that we do not over consume and yet at the same time, look to go to Mars and beyond. Hopefully, we spend more time with our families. Hopefully, we take care of our health more. Hopefully, we care less for the glossy social media posts of celebrities, we do not know and care more for the people we know. Hopefully, we wise up and spend more time figuring out why are we here. It is not about “I” or my family but as they say it’s us and our family as a country and world that matters. This war cannot be won by technology or military might but by people’s resolve and restrain. So, let us stand up and make way for the future generations. We are in war with an invisible virus and war with self to show the commitment and build what could be a “Vasudeva Kutumbakam”. It is not important we do our best. Sometimes we must do what is required.
The future of elective surgeries in the ‘new normal’
Jun 5, 2020 (1 min read)Join this interactive webinar with DR. RAVI SAMY, DR. JAMES NAPLES, and DR. SCOTT SHAPIRO from UC along with DR. SAMPATH CHANDRA PRASAD...
 Dr.
Sampath Chandra Prasad Rao
Dr.
Sampath Chandra Prasad Rao
The future of elective surgeries in the ‘new normal’
Join this interactive webinar with DR. RAVI SAMY, DR. JAMES NAPLES, and DR. SCOTT SHAPIRO from UC along with DR. SAMPATH CHANDRA PRASAD RAO from MANIPAL HOSPITAL, BANGALORE and PROF. PER CAYE THOMASEN, from ORL-HNS UNIVERSITY CLINIC, COPENHAGEN as they discuss their thoughts on the future of surgery after the pandemic.
Date Time: May 1, 2020 09:00 AM Eastern Time (US and Canada)
The panel will discuss topics such as decision-making, the state of elective surgery, telemedicine, and many others. This webinar is the third installation of a series on surgery in the COVID 19 era, brought to you by CSurgeries. To access the previous recordings please go to: www.csurgeries.com

Hearing restoration through cochlear & other implants (for public)
May 9, 2020 (1 min read)Hearing is one of the most essential senses, and losing it can turn a person's life upside down. To help you understand how it can be...
 Dr.
Sampath Chandra Prasad Rao
Dr.
Sampath Chandra Prasad Rao
Hearing restoration through cochlear & other implants (for public)

Hearing is one of the most essential senses, and losing it can turn a person's life upside down. To help you understand how it can be restored, Dr Sampath Chandra Prasad Rao will be joining us in a Facebook Live video session on 18th April, 10:00 AM to 10:30 AM. #ApolloSpectra
Date Time: May 1, 2020 09:00 AM Eastern Time (US and Canada)
The panel will discuss topics such as decision-making, the state of elective surgery, telemedicine, and many others. This webinar is the third installation of a series on surgery in the COVID 19 era, brought to you by CSurgeries. To access the previous recordings please go to: www.csurgeries.com


Coronavirus disease (COVID-19) advice for the public by World Health Organisation
Mar 13, 2020 (3 min read)Stay aware of the latest information on the COVID-19 outbreak, available on the WHO website and through your national and local public...
 Shamili Asokan
Shamili Asokan
Coronavirus disease (COVID-19) advice for the public by World Health Organisation
Stay aware of the latest information on the COVID-19 outbreak, available on the WHO website and through your national and local public health authority. COVID-19 is still affecting mostly people in China with some outbreaks in other countries. Most people who become infected experience mild illness and recover, but it can be more severe for others. Take care of your health and protect others by doing the following: Wash your hands frequently Regularly and thoroughly clean your hands with an alcohol-based hand rub or wash them with soap and water. Why? Washing your hands with soap and water or using alcohol-based hand rub kills viruses that may be on your hands. Maintain social distancing Maintain at least 1 metre (3 feet) distance between yourself and anyone who is coughing or sneezing. Why? When someone coughs or sneezes they spray small liquid droplets from their nose or mouth which may contain virus. If you are too close, you can breathe in the droplets, including the COVID-19 virus if the person coughing has the disease. Avoid touching eyes, nose and mouth Why? Hands touch many surfaces and can pick up viruses. Once contaminated, hands can transfer the virus to your eyes, nose or mouth. From there, the virus can enter your body and can make you sick. Practice respiratory hygiene Make sure you, and the people around you, follow good respiratory hygiene. This means covering your mouth and nose with your bent elbow or tissue when you cough or sneeze. Then dispose of the used tissue immediately. Why? Droplets spread virus. By following good respiratory hygiene you protect the people around you from viruses such as cold, flu and COVID-19. If you have fever, cough and difficulty breathing, seek medical care early Stay home if you feel unwell. If you have a fever, cough and difficulty breathing, seek medical attention and call in advance. Follow the directions of your local health authority. Why? National and local authorities will have the most up to date information on the situation in your area. Calling in advance will allow your health care provider to quickly direct you to the right health facility. This will also protect you and help prevent spread of viruses and other infections. Stay informed and follow advice given by your healthcare provider Stay informed on the latest developments about COVID-19. Follow advice given by your healthcare provider, your national and local public health authority or your employer on how to protect yourself and others from COVID-19. Why? National and local authorities will have the most up to date information on whether COVID-19 is spreading in your area. They are best placed to advise on what people in your area should be doing to protect themselves. Protection measures for persons who are in or have recently visited (past 14 days) areas where COVID-19 is spreading Follow the guidance outlined above. Stay at home if you begin to feel unwell, even with mild symptoms such as headache and slight runny nose, until you recover. Why? Avoiding contact with others and visits to medical facilities will allow these facilities to operate more effectively and help protect you and others from possible COVID-19 and other viruses. If you develop fever, cough and difficulty breathing, seek medical advice promptly as this may be due to a respiratory infection or other serious condition. Call in advance and tell your provider of any recent travel or contact with travelers. Why? Calling in advance will allow your health care provider to quickly direct you to the right health facility. This will also help to prevent possible spread of COVID-19 and other viruses. #medicalschool #tips #advice

Two day workshop with live surgeries to demonstrate complex skull based procedures held at
Feb 7, 2020 (2 min read)World Skull Base and Columbia Asia Hospital, Sarjapur Road conducted a two-day live workshop to telecast complex skull based surgeries to...
 Shamili Asokan
Shamili Asokan
Two day workshop with live surgeries to demonstrate complex skull based procedures held at CAHSR

World Skull Base and Columbia Asia Hospital, Sarjapur Road conducted a two-day live workshop to telecast complex skull based surgeries to ENT surgeons and Neuro Surgeons across Karnataka & Andhra Pradesh. In the two-day event, the participating surgeons attended series of lectures, witnessed cadaver dissection and live telecast of 3 complex base of skull surgeries in the hospital.Around 75 ENT surgeons, Neuro surgeons and Radiologists across Karnataka and Andhra Pradesh attended the workshop. Additionally, 700 doctors witnessed the online live telecast proceedings through various media handles.The programme focused on Base of Skull surgeries: Lectures included the anatomical, radiological and surgical aspects of Skull Base Surgery. They also highlighted the support systems and recent advances such as Neuro-navigation, neuro-monitoring and neuro-anaesthesia and hearing rehabilitation solutions for deafness during pathology like Bone Anchored hearing Implants.Companies like Cochlear (for BAHA), Sanma (for microscopes) and Medtronic and Bien Air (for drill systems) supported the grand event.One of the key highlights of the first day of the event was a six hours’ marathon on cadaveric demonstration of multiple Skull Base procedures by Dr. Sampath Chandra Prasad Rao and Dr. Manjunath Dandinarasaiah. The second day of the event was live surgery demonstration of complex skull based procedures such as Petrous Bone Cholesteatoma, Skull Base Osteomyelitis, Nerve grafting for Facial Nerve repair and Translabyrinthine Approach for Vestibular Schwannoma.Dr. Sampath Rao is an internationally acclaimed Skull Base Surgeon working at Columbia Asia Hospital Sarjapur Road. He has won several awards and has been invited for more than 200 important academic events all across the world. He has more than 100 peer reviewed publications on the subject.Speaking about the event, Ms Manisha Kumar, General Manager of the hospital said “We are delighted to be one of the pioneers in the field of academics and continuous learning in the Skull Based surgery domain. It has been our endeavour to build centres of excellence in super specialties and this workshop is another step towards connecting with the clinical fraternity in establishing base of skull surgeries as a super specialty program.”